ICSE Class 10 Answered
can you please suggest me a teacher who can explain me mole concept perfectly!!!!!
Asked by pawarkrithika1234 | 11 Mar, 2019, 09:14: PM
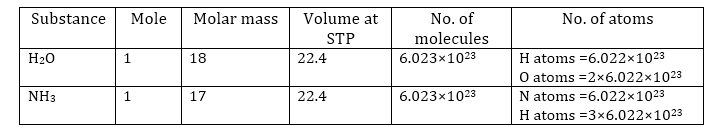
Mole Concept:
1 mole of a substance is equal to its atomic mass or molecular mass expressed in grams.
The atomic mass of sodium is 23 grams.
Therefore, 23 grams of sodium is equal to one mole of sodium atoms.
Similarly, the molecular mass of oxygen (O2) = 2 × Atomic mass of oxygen
= 2 × 16 = 32 g
So, 32 grams of oxygen is equal to one mole of oxygen molecules.
1 mole (of anything) = 6.022 × 1023 in number
For questions based on mole concept please refer the below link:
Answered by Varsha | 12 Mar, 2019, 10:58: AM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by jrvedant208 | 05 Feb, 2024, 10:37: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 10 Jul, 2022, 10:13: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 25 Jun, 2022, 10:24: PM
ICSE 10 - Chemistry
Asked by palshivom72 | 14 Jul, 2020, 07:56: PM
ICSE 10 - Chemistry
Asked by jhabijay01 | 27 May, 2020, 12:20: PM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:53: AM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:37: AM
ICSE 10 - Chemistry
Asked by aashimegh | 28 Aug, 2019, 05:25: PM