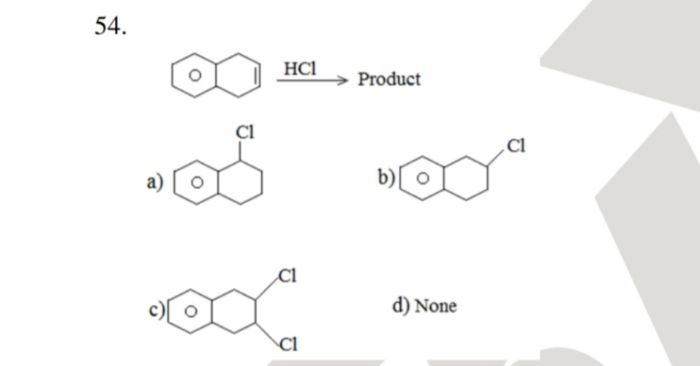
CBSE Class 12-science Answered
10.18 ques plz

Asked by lovemaan5500 | 31 Dec, 2019, 04:58: PM
When Alkyl chloride is treated with aq. KOH- It is a nucleophilic substitution reaction. KOH releases OH- ion in aquous medium and this OH- ion replaces Cl- and alcohol is formed.
RCl + aq. KOH --------> ROH + KCl
When Alkyl chloride is treated with alcoholic KOH- It is a beta-elimination reaction. Alcoholic KOH produces alkoxide ion (RO_) ion which is a strong base, it removes hydrogen from beta-Carbon of alkyl chloride and alkene is formed.
Answered by Ravi | 31 Dec, 2019, 06:00: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by jaiadithya05 | 20 Sep, 2021, 07:20: PM
CBSE 12-science - Chemistry
Asked by shantasharma611 | 01 May, 2021, 01:51: PM
CBSE 12-science - Chemistry
Asked by sivaveeramachaneni9 | 08 Feb, 2021, 09:47: PM
CBSE 12-science - Chemistry
Asked by kalkikai33 | 01 Jun, 2020, 03:40: PM
CBSE 12-science - Chemistry
Asked by subhasmitaswainstudent | 02 May, 2020, 01:22: PM
CBSE 12-science - Chemistry
Asked by buluacharya123 | 25 Apr, 2020, 11:37: AM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 14 Apr, 2020, 10:58: PM
CBSE 12-science - Chemistry
Asked by tn6380313887.mohanviji | 09 Mar, 2020, 09:19: AM
CBSE 12-science - Chemistry
Asked by nidhi.jain0212 | 07 Mar, 2020, 01:30: PM