CBSE Class 11-science - Balancing Redox Reactions Videos
Redox Reactions
This video explains the half reaction method and the half reaction method to balance redox reactions in acidic and basic media.
More videos from this chapter
View All- what is the most essential conditions that must be satisfied in a redox reaction
- H2O2+Fe2+=H2O +Fe3+ , give ion exchange method
-
Redox Reaction: solve the following equation by ion electron method in acidic medium
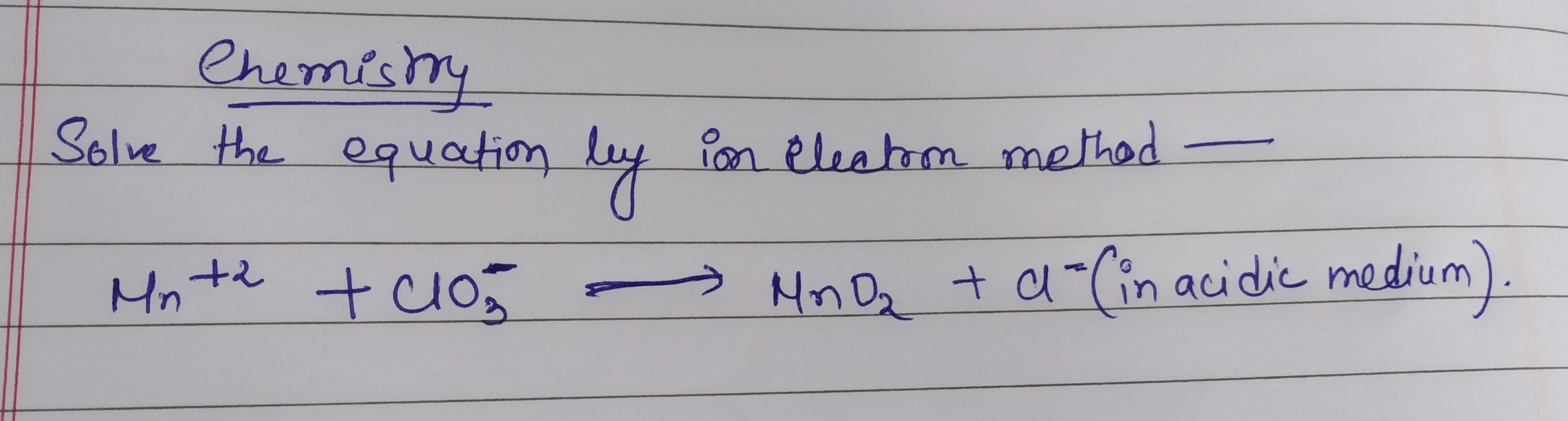
- NO3 (-ve)+I (-ve)+H (+) =NO +I2 +H2O
- magnesium reacts with nitric acid to give magnesium nitarate and nitrous oxide gas and liquid water balance this by oxidation number method
- H2S + KMnO4 + H2SO4 → S+ MnSO4 + KHSO4 + H2O Balance by oxidation number method step by step explain please
- Zn + HNO3 = Zn(NO3) + NO2 + H2O balance this equation by oxidation number method step wise
- MO4 minus + I gives MnO2+I2
-
Solve it

- Define oxidation in terms of oxidation numbers.






