CBSE Class 12-science Answered
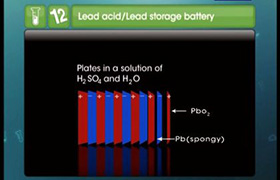
Write the cell reactions which occur in lead storage battery (i) when the battery is in use (ii) when the battery is on charging.
Asked by Topperlearning User | 22 Jun, 2016, 14:58: PM
At Anode : Pb (s) + SO4 2- (aq) → PbSO4 (s)+ 2e-.
At Cathode: PbO2(s) + SO4 2- (aq) + 4H+ (aq) +2e- →PbSO4 (s) + 2H2O(l)
The overall reaction is
Pb (s) + PbO2(s) + 4H+ (aq) + 2SO4 2-(aq) → 2PbSO4 (s) + 2H2O(l)
While charging:
At Cathode: PbSO4 (s)+ 2e- → Pb (s) + SO4 2- (aq)
At Anode: PbSO4 (s) + 2H2O(l) → PbO2(s) + SO4 2- (aq) + 4H+ (aq) +2e-
Answered by | 22 Jun, 2016, 16:58: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by Topperlearning User | 16 Apr, 2014, 10:35: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 14:58: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 14:53: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 22 Jun, 2016, 14:53: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM