CBSE Class 11-science Answered
Which should have a lower boiling point H2O or H2S?
Asked by Topperlearning User | 17 Jun, 2016, 11:23: AM
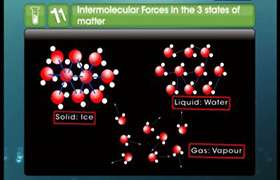
H2S have a lower boiling point i.e. -70oC because no hydrogen bonding is present in liquid H2S whereas the boiling point of water is 100oC due to the presence of very strong hydrogen bonding.
Answered by | 17 Jun, 2016, 01:23: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by luvs6482 | 27 Apr, 2024, 08:18: PM
CBSE 11-science - Chemistry
Asked by smanishkumar2002 | 04 Aug, 2018, 05:36: AM
CBSE 11-science - Chemistry
Asked by tps.mjmdr | 22 Jun, 2018, 06:36: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 02:31: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 02:33: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 02:34: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 02:36: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 17 Jun, 2016, 11:24: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 17 Jun, 2016, 11:23: AM