CBSE Class 11-science Answered
Which has a higher boiling point I2 or Br2?
Asked by Topperlearning User | 17 Jun, 2016, 11:24: AM
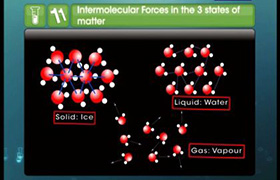
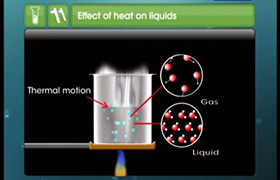
I2 has a higher boiling point than Br2. The atomic weights of Br and I are 80 and 127 respectively. Since I2 has higher molecular weight, it has stronger London dispersion forces so it has a higher boiling point than Br2.
Answered by | 17 Jun, 2016, 13:24: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by luvs6482 | 27 Apr, 2024, 20:18: PM
CBSE 11-science - Chemistry
Asked by smanishkumar2002 | 04 Aug, 2018, 05:36: AM
CBSE 11-science - Chemistry
Asked by tps.mjmdr | 22 Jun, 2018, 18:36: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 14:31: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 14:33: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 14:34: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 20 Apr, 2015, 14:36: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 17 Jun, 2016, 11:24: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 17 Jun, 2016, 11:23: AM