CBSE Class 12-science Answered
What is the difference between catalyst and catalysis?
Asked by Topperlearning User | 21 Jun, 2016, 12:07: PM
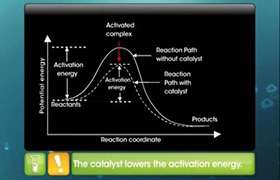
A catalyst is defined as a substance, which accelerates the rate of a chemical reaction and is itself unchanged and not consumed in the overall reaction. Catalysis is the phenomenon of altering the rate of a reaction with the help of a catalyst.
Answered by | 21 Jun, 2016, 02:07: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by sivakapuganti1 | 26 Aug, 2020, 08:57: PM
CBSE 12-science - Chemistry
Asked by spoorthysaienelluri | 23 May, 2020, 11:07: AM
CBSE 12-science - Chemistry
Asked by vermahitesh124 | 12 May, 2020, 07:58: AM
CBSE 12-science - Chemistry
Asked by yogeshsulakh | 07 Feb, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by tlb2bpartner | 21 Aug, 2019, 10:28: PM
CBSE 12-science - Chemistry
Asked by niharikapabba2605 | 07 Aug, 2018, 11:38: AM
CBSE 12-science - Chemistry
Asked by pardeepkumar2281 | 31 Jul, 2018, 11:40: PM
CBSE 12-science - Chemistry
Asked by pardeepkumar2281 | 31 Jul, 2018, 11:38: PM
CBSE 12-science - Chemistry
Asked by pardeepkumar2281 | 31 Jul, 2018, 11:36: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM