CBSE Class 12-science Answered
what is mean by +I inductive effect and why it is responsible for basicity of amines?
Asked by | 01 Apr, 2013, 10:44: PM
Inductive effect is the process of electron displacement of electrons along the chain of carbon atoms due to the presence of a polar covalent bond at one end of the chain. Atoms or groups which push electron density away from themselves exhibit positive Inductive effect (+I). These atoms or groups have a greater electron releasing power than hydrogen.
Answered by | 02 Apr, 2013, 11:03: AM
Concept Videos
CBSE 12-science - Chemistry
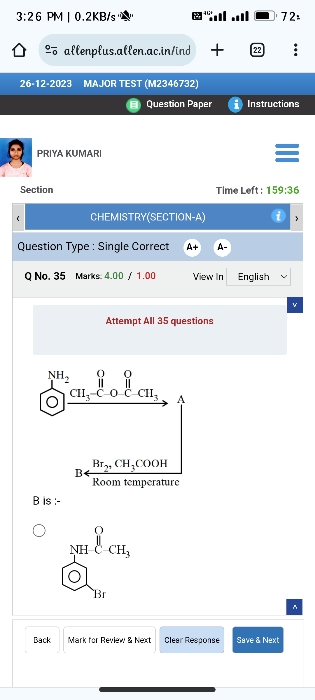
Asked by hanihope27 | 01 Mar, 2024, 08:33: PM
CBSE 12-science - Chemistry
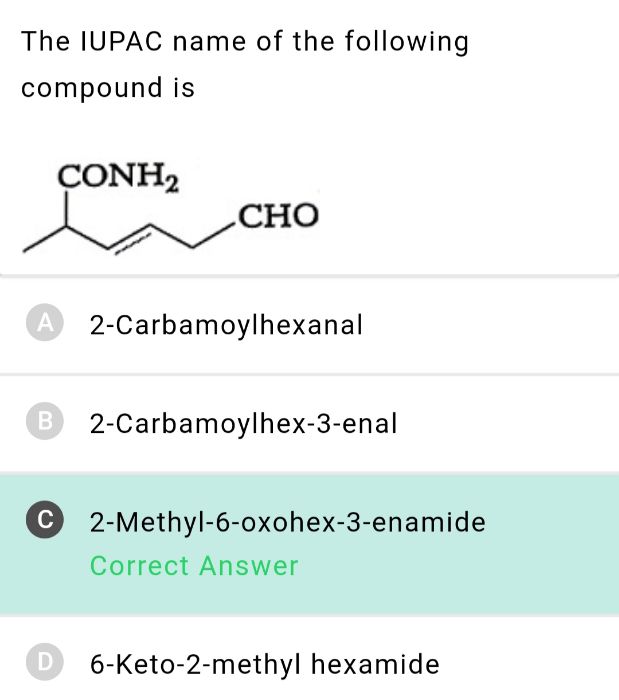
Asked by priyankapaliwal255 | 23 Sep, 2023, 05:46: AM
CBSE 12-science - Chemistry
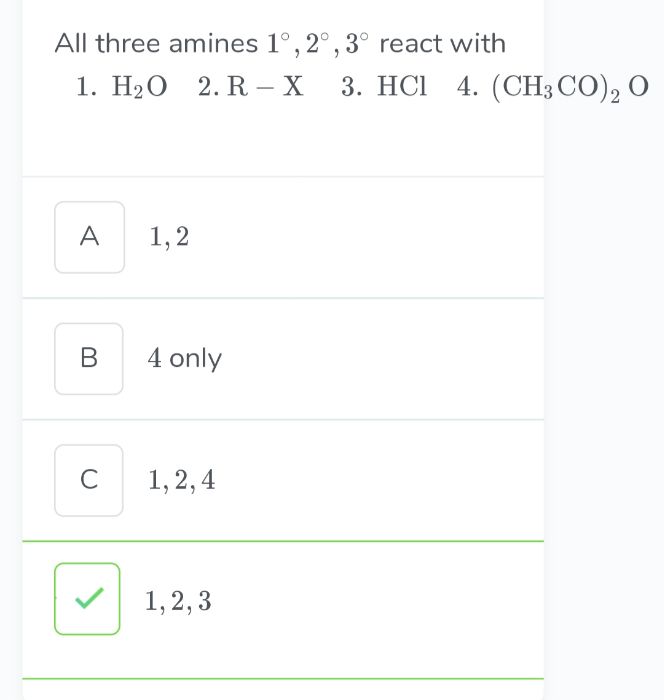
Asked by shwetayaligar205 | 07 Jul, 2022, 08:13: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 06 Jul, 2021, 11:31: PM
CBSE 12-science - Chemistry
Asked by dhivagar25375 | 12 Aug, 2020, 08:34: PM
CBSE 12-science - Chemistry
Asked by danapalanandhan | 28 Jul, 2020, 11:48: AM
CBSE 12-science - Chemistry
Asked by sulaikhasulu393 | 27 May, 2020, 03:34: PM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 15 Apr, 2020, 01:35: PM