CBSE Class 11-science Answered
What is bond pair and lone pair of electrons?
Asked by Topperlearning User | 08 Oct, 2014, 08:51: AM
Bond pair of electrons is the electron pair that is directly involved in bond formation. The lone pair of electrons is the electron pair which is not involved directly in bonding.
Answered by | 08 Oct, 2014, 10:51: AM
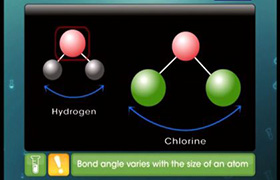
Concept Videos
CBSE 11-science - Chemistry
Asked by rushikeshpandav00 | 17 Jun, 2021, 13:43: PM
CBSE 11-science - Chemistry
Asked by mittalneetu345 | 12 Feb, 2021, 10:23: AM
CBSE 11-science - Chemistry
Asked by abdulhamid02427 | 24 Dec, 2020, 11:51: AM
CBSE 11-science - Chemistry
Asked by rayyan20151 | 09 Jan, 2020, 11:40: AM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 07 Sep, 2019, 15:59: PM
CBSE 11-science - Chemistry
Asked by narayandhar1979 | 28 Oct, 2018, 10:25: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 08 Oct, 2014, 08:44: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 07 Oct, 2014, 17:14: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 08 Oct, 2014, 08:51: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM