CBSE Class 11-science Answered
The de broglie wavelength of a particle with mass 1kg and velocity 100m/s is
Asked by arushidabhade | 08 Jul, 2019, 20:05: PM
Given:
mass, m = 1 kg
velocity, v = 100 m/s
h = 6.63×10-27 erg.sec
De broglie wavelength is given by,
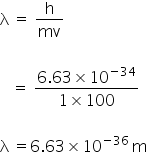
De Brogli's wavelength is 6.63×10-36 m
Answered by Varsha | 09 Jul, 2019, 14:52: PM
Concept Videos
CBSE 11-science - Chemistry
The de Broglie wavelength of an electron travelling with 20% of velocity of light is nearly equal to
Asked by arushidabhade | 13 Aug, 2019, 13:32: PM
CBSE 11-science - Chemistry
Asked by arushidabhade | 13 Aug, 2019, 13:31: PM
CBSE 11-science - Chemistry
Asked by arushidabhade | 13 Aug, 2019, 13:14: PM
CBSE 11-science - Chemistry
Asked by arushidabhade | 08 Jul, 2019, 20:07: PM
CBSE 11-science - Chemistry
Asked by arushidabhade | 08 Jul, 2019, 20:05: PM
CBSE 11-science - Chemistry
Asked by bjayanta | 15 Apr, 2019, 10:39: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 13:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Apr, 2015, 18:22: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Apr, 2015, 18:32: PM