CBSE Class 12-science Answered
sir/madam,
in NCERT chemistry books in organic chemistry sometimes hydrolysis rxn takes places by acid and water(H2SO4 + H2O/ H3O+/H+ + H2)), base with water(NaOH + H2O/ KOH + H2O) and other times only with water(H2O)
what is the difference between the three and when do we use them in rxns ?
hydrolysis means breaking or cleavage of compounds using water.... am I right?
request u to answer all questions pls
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
Hydrolysis means reaction of organic compound with water to form two or more new substances. In hydrolysis reaction, Cleavage of bond take place and water is added.
In different organic compounds cleavage/breaking of bond takes place in different ways so for that purpose we use different reagents. But for addition Only H2O is required so H2O is common in all of them.
Answered by Ravi | 01 Jul, 2021, 10:21: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
CBSE 12-science - Chemistry
Asked by mahaynoorf | 17 Oct, 2020, 20:39: PM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 14 Apr, 2020, 22:58: PM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 14 Apr, 2020, 11:54: AM
CBSE 12-science - Chemistry
Asked by govtsecschoolnayaganv051 | 28 Aug, 2019, 19:45: PM
CBSE 12-science - Chemistry
Asked by dineshchem108 | 20 May, 2019, 23:46: PM
CBSE 12-science - Chemistry
Asked by shobhit | 21 Feb, 2019, 23:01: PM
CBSE 12-science - Chemistry
Asked by Atulcaald | 16 May, 2018, 14:43: PM
CBSE 12-science - Chemistry
Asked by Atulcaald | 16 May, 2018, 14:41: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 08 Apr, 2014, 08:33: AM




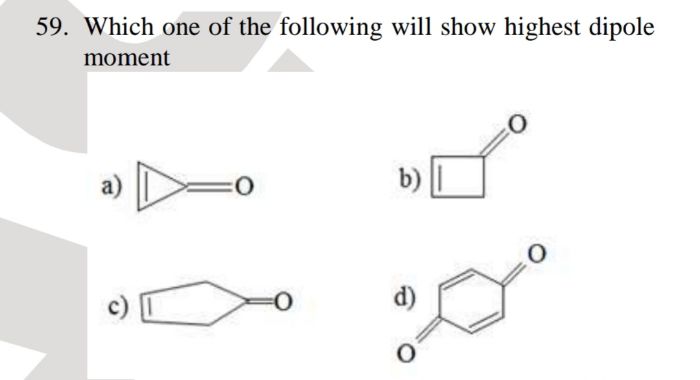
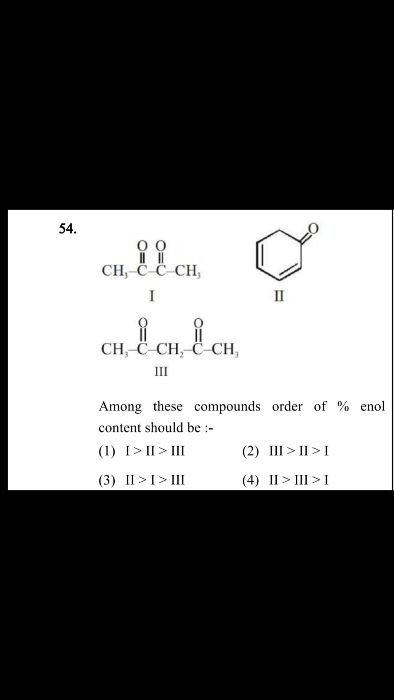
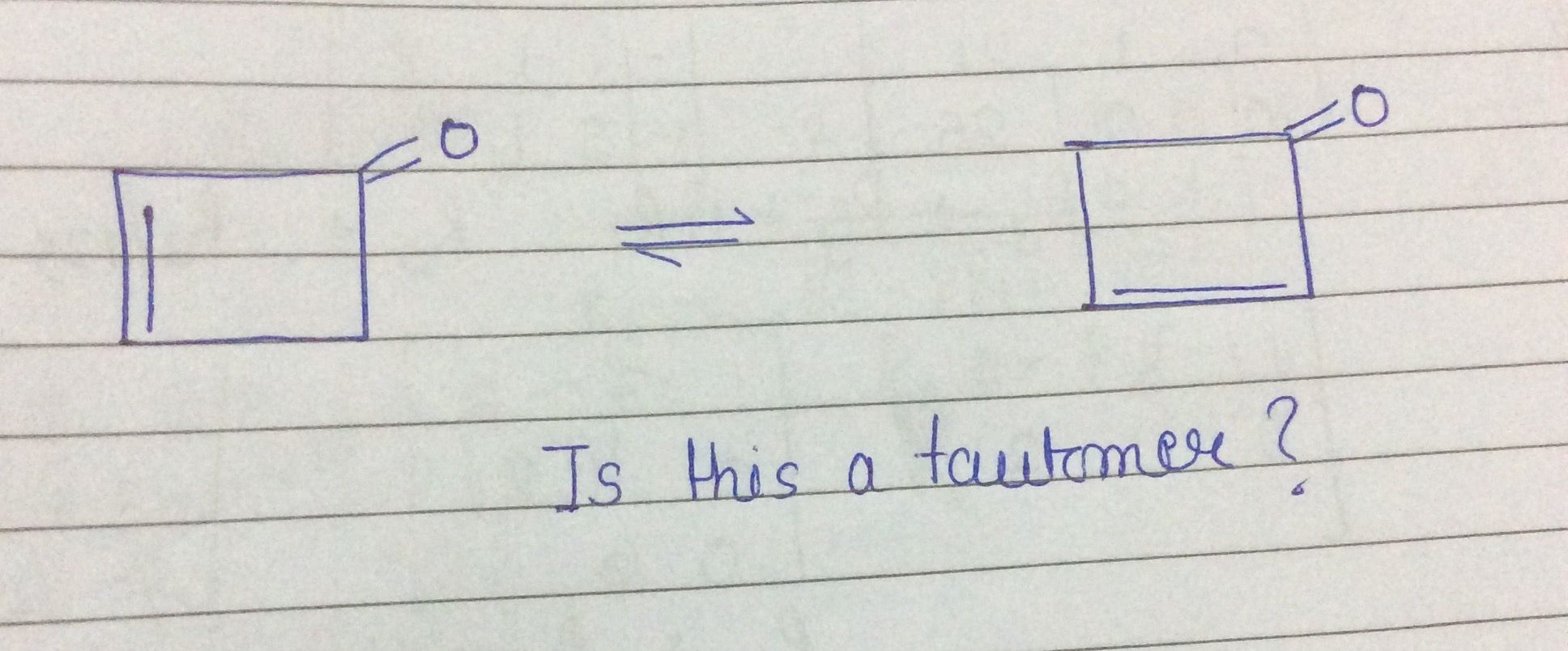
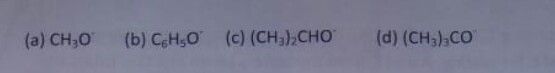
 Y reacts with Z to give
(1)
Y reacts with Z to give
(1)
 (2)
(2)
 (3)
(3)
 (4)
(4)

 Z is :-
(1)
Z is :-
(1)
 (2)
(2)
 (3)
(3)
 (4)
(4)

