CBSE Class 12-science Answered
Q=with respect to aldehydes ,ketones of comparable molecular masses have a higher boiling point than aldehydes.Explain why is it so?
Asked by vaibhav gupta | 02 Jan, 2014, 11:49: AM
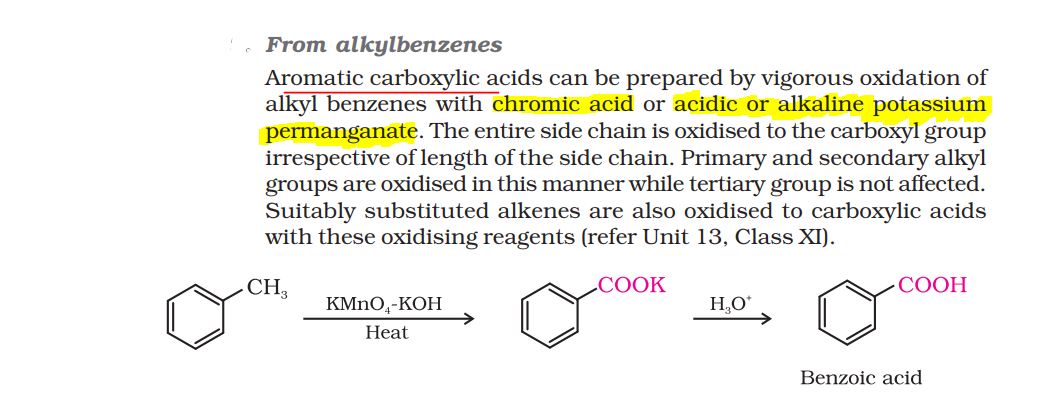
For ketones and aldehydes of similar molecular mass, ketones have higher boiling point.
This is due to the fact that its carbonyl group in ketone is more polarized than in aldehydes.
The presence of two electron donating alkyl groups around the C=O group in ketone makes the molecule more polar than in case of aldehydes.
Hence, the interactions between molecules of ketones is stronger than between molecules of aldehydes, which results into a higher boiling point of ketone to that of corresponding aldehyde of similar mass.
Answered by Prachi Sawant | 02 Jan, 2014, 01:05: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by ukg8612 | 15 Apr, 2024, 07:36: PM
CBSE 12-science - Chemistry
Asked by ajayarchi | 08 Feb, 2024, 03:43: AM
CBSE 12-science - Chemistry
Asked by pallasriramulu9 | 24 Dec, 2023, 06:05: AM
CBSE 12-science - Chemistry
Asked by bsaheliya | 22 Dec, 2023, 09:53: PM
CBSE 12-science - Chemistry
Asked by ygarg8323 | 18 Apr, 2022, 12:47: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 30 Jun, 2021, 04:52: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 28 Jun, 2021, 02:34: PM
CBSE 12-science - Chemistry
Asked by saimerala007 | 22 May, 2021, 02:08: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 31 Dec, 2020, 10:45: AM