CBSE Class 12-science Answered
sir/madam,
is KMnO4-KOH the chemical formula for alkaline potassium permanganate?
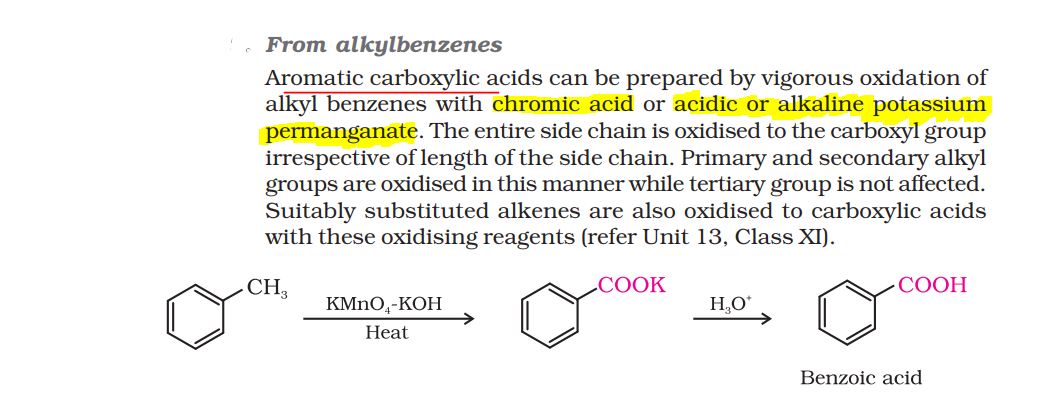
like in the NCERT if alkylbenzene is reacted with chromic acid (H2CrO4), will there be any intermediate product just like alkylbenzene when reacted with KMnO4-KOH gives potassium salt of carboxylic acid which then further oxidises to carboxylic acid?
also is my formula correct for chromic acid and alkaline potassium permanganate?
pls requested to respond to all my questions
Asked by kaziryan.05 | 28 Jun, 2021, 14:34: PM
Dear Student,
Alkalis are bases which are soluble in water. Example of alkalis are- NaOH, KOH etc.
So, Alkali potasssium permagnate means- NaOH/KOH with KMnO4 . So mentioned formula is correct.
if alkylbenzene is reacted with chromic acid, there be ntermediate but that mechanism is complex and not the part of syllabus. We just have to remember end proiduct.
Formula of chromic acid- H2CrO4
Answered by Ravi | 28 Jun, 2021, 17:05: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by ankitmonda.bankhatil | 11 Jun, 2024, 19:10: PM
CBSE 12-science - Chemistry
Asked by ukg8612 | 15 Apr, 2024, 19:36: PM
CBSE 12-science - Chemistry
Asked by ajayarchi | 08 Feb, 2024, 03:43: AM
CBSE 12-science - Chemistry
Asked by pallasriramulu9 | 24 Dec, 2023, 06:05: AM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 30 Jun, 2021, 16:52: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 28 Jun, 2021, 14:34: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 31 Dec, 2020, 10:45: AM
CBSE 12-science - Chemistry
Asked by shreevarshni1910 | 16 Jun, 2020, 13:36: PM
CBSE 12-science - Chemistry
Asked by yukthas706 | 11 Jun, 2020, 10:00: AM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 14 Feb, 2020, 12:09: PM