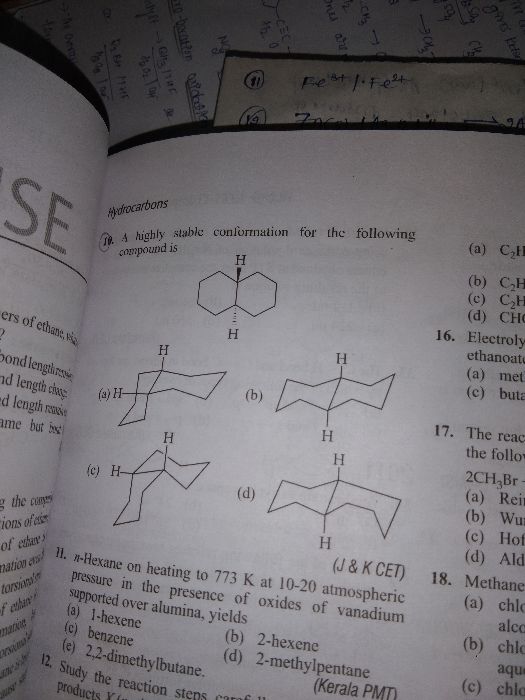
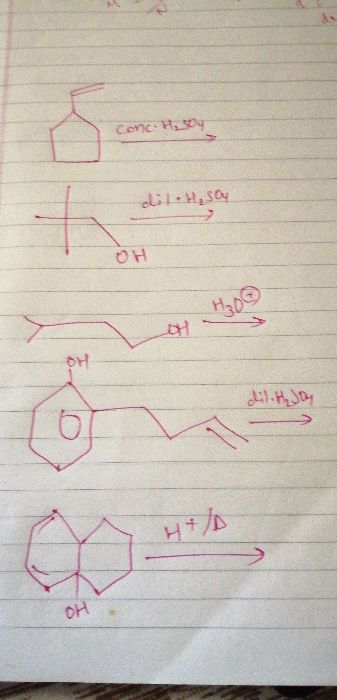
CBSE Class 11-science Answered
Out of alkanes, alkenes and alkynes....the energy required to break the triple bond is the greatest!.. then which of them is the strongest and the most stable?
Asked by Meghna G. | 21 Nov, 2012, 06:07: PM
Out of alkanes, alkenes and alkynes; alkynes are the most strongest since they contain C C bond; strength of C
C bond; strength of C C bond is more than that of C=C double bond and C-C single bond. So, these are the most stable and strong as compared to alkenes and alkanes. Alkanes contain weak covalent bond binded by weak vander waal forces whereas alkenes are very reactive because of presence of
C bond is more than that of C=C double bond and C-C single bond. So, these are the most stable and strong as compared to alkenes and alkanes. Alkanes contain weak covalent bond binded by weak vander waal forces whereas alkenes are very reactive because of presence of  (pi) bond.
(pi) bond.
 C bond; strength of C
C bond; strength of C C bond is more than that of C=C double bond and C-C single bond. So, these are the most stable and strong as compared to alkenes and alkanes. Alkanes contain weak covalent bond binded by weak vander waal forces whereas alkenes are very reactive because of presence of
C bond is more than that of C=C double bond and C-C single bond. So, these are the most stable and strong as compared to alkenes and alkanes. Alkanes contain weak covalent bond binded by weak vander waal forces whereas alkenes are very reactive because of presence of  (pi) bond.
(pi) bond.
Answered by | 23 Nov, 2012, 10:39: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by kirithshiv | 24 Feb, 2024, 12:12: PM
CBSE 11-science - Chemistry
Asked by shivanij4734 | 16 Dec, 2023, 08:30: PM
CBSE 11-science - Chemistry
Asked by maibamjohnny89 | 15 Jan, 2022, 09:38: PM
CBSE 11-science - Chemistry
Asked by archu312004 | 07 Feb, 2021, 10:21: PM
CBSE 11-science - Chemistry
Asked by dubeyanubhav65 | 18 Jan, 2021, 09:52: PM
CBSE 11-science - Chemistry
Asked by shahsaqlain107 | 30 Nov, 2020, 01:10: PM
CBSE 11-science - Chemistry
Asked by ghastipratiksha | 11 Jul, 2020, 08:14: PM
CBSE 11-science - Chemistry
Asked by guptaserendri | 01 Jul, 2020, 03:58: PM
CBSE 11-science - Chemistry
Asked by Manpreetsingh669933 | 13 Apr, 2020, 01:39: PM
CBSE 11-science - Chemistry
Asked by bittutiwary1234 | 23 Feb, 2020, 12:15: AM