CBSE Class 12-science Answered
name the two groups into which phenomenon of catalysts can be divided give an example of each group with chemical equation involved
Asked by niharikapabba2605 | 07 Aug, 2018, 11:38: AM
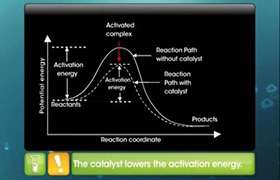
The phenomenon of catalysis is divided into two groups:
Types of Catalysis
(A) Homogeneous Catalysis
In this, both the catalysts and the reactants are in the same phase.
Example,(i) Catalytic oxidation of SO2 into SO3 in the presence of NO as a catalyst in the lead chamber process. In this case, all the reactant and the catalyst are in the gaseous phase.

(ii) Hydrolysis of ethyl acetate in the presence of the acid

(B) Heterogeneous Catalysis
In this, both the catalysts and the reactant are in different phase.
Example: Some common examples of heterogeneous catalysis are as follows.
(i) Oxidation of HCl into Cl2 by Deason’s process in the presence of CuCl2.
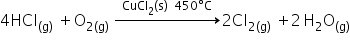
(ii) Manufacture of NH3 from N2 and H2 by Haber’s process using finely divided iron as the catalyst.
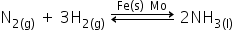
Answered by Ramandeep | 07 Aug, 2018, 04:09: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by sivakapuganti1 | 26 Aug, 2020, 08:57: PM
CBSE 12-science - Chemistry
Asked by spoorthysaienelluri | 23 May, 2020, 11:07: AM
CBSE 12-science - Chemistry
Asked by vermahitesh124 | 12 May, 2020, 07:58: AM
CBSE 12-science - Chemistry
Asked by yogeshsulakh | 07 Feb, 2020, 09:28: AM
CBSE 12-science - Chemistry
Asked by tlb2bpartner | 21 Aug, 2019, 10:28: PM
CBSE 12-science - Chemistry
Asked by niharikapabba2605 | 07 Aug, 2018, 11:38: AM
CBSE 12-science - Chemistry
Asked by pardeepkumar2281 | 31 Jul, 2018, 11:40: PM
CBSE 12-science - Chemistry
Asked by pardeepkumar2281 | 31 Jul, 2018, 11:38: PM
CBSE 12-science - Chemistry
Asked by pardeepkumar2281 | 31 Jul, 2018, 11:36: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM