CBSE Class 9 Answered
in a reaction 4.0g of sodium carbonate were reacted with 10g of hydrochloric acid. The product was a mixture of 2.5g of carbon dioxide and 11.5g of sodium chloride.

Asked by rjinaaishu007 | 10 Feb, 2020, 19:05: PM
The reaction is:
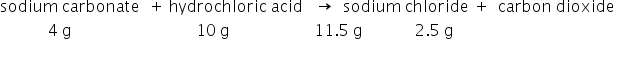
Mass of reactants = 4+10 = 14 gm
Mass of products = 11.5 + 2.5 = 14 gm
The mass of reactants is equal to the mass of the products. Therefore the given data is in agreement with law of conservation of mass.
Answered by Varsha | 11 Feb, 2020, 11:29: AM
Application Videos
Concept Videos
CBSE 9 - Chemistry
Asked by Niharikadhamija13 | 25 Aug, 2020, 17:17: PM
CBSE 9 - Chemistry
Asked by haritchahar | 25 Jul, 2020, 11:55: AM
CBSE 9 - Chemistry
Asked by yanagauswami00.tl | 17 Apr, 2020, 10:44: AM
CBSE 9 - Chemistry
Asked by harshilmodi74.tl | 16 Apr, 2020, 10:39: AM
CBSE 9 - Chemistry
Asked by harshilmodi74.tl | 16 Apr, 2020, 10:35: AM
CBSE 9 - Chemistry
Asked by rjinaaishu007 | 10 Feb, 2020, 19:05: PM
CBSE 9 - Chemistry
Asked by prakash.sanyasi | 09 Feb, 2020, 22:59: PM
CBSE 9 - Chemistry
Asked by kumaruditanshu27 | 21 Oct, 2019, 18:16: PM
CBSE 9 - Chemistry
Asked by guptarushil6 | 15 Oct, 2019, 22:41: PM
CBSE 9 - Chemistry
Asked by lopamudrabasak1996 | 11 Aug, 2019, 00:51: AM







