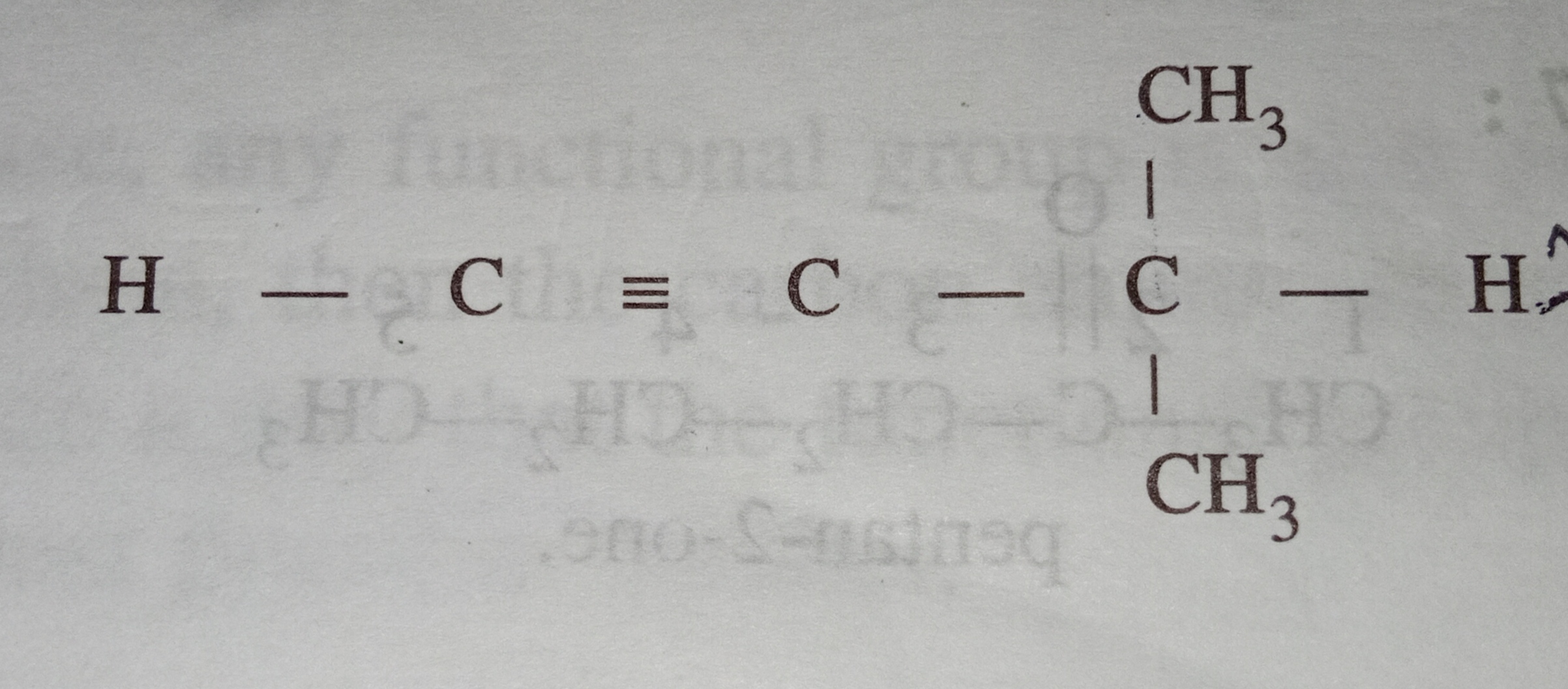CBSE Class 11-science Answered
if the carbon bonded to halogen is  hybridised ,how will it be classified?
hybridised ,how will it be classified?
 hybridised ,how will it be classified?
hybridised ,how will it be classified?
Asked by ABHILASHA | 22 Aug, 2020, 05:09: AM
if the carbon bonded to halogen is sp2 hybridised carbon of alkene then it will be known as allylic halide.
If the carbon bonded to halogen is sp2 hybridised carbon of benzene then it will be known as aryl halide.
Answered by Ravi | 22 Aug, 2020, 05:55: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by neet2025targetgo | 25 Mar, 2024, 10:13: AM
CBSE 11-science - Chemistry
Asked by pamjat.8888 | 31 Jan, 2024, 11:31: AM
CBSE 11-science - Chemistry
Asked by sahumahesh3973 | 20 Jan, 2024, 06:33: PM
CBSE 11-science - Chemistry
Asked by aswintj2007 | 07 Jan, 2024, 08:53: PM
CBSE 11-science - Chemistry
Asked by dipalisingh0908 | 05 Nov, 2023, 02:24: PM
CBSE 11-science - Chemistry
Asked by badalbehera258369 | 19 Oct, 2023, 02:01: PM
CBSE 11-science - Chemistry
Asked by prakrutikhosla | 16 Sep, 2023, 06:31: PM
CBSE 11-science - Chemistry
Asked by hitanshu04 | 17 Jun, 2021, 07:20: PM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 22 Aug, 2020, 05:13: AM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 22 Aug, 2020, 05:09: AM