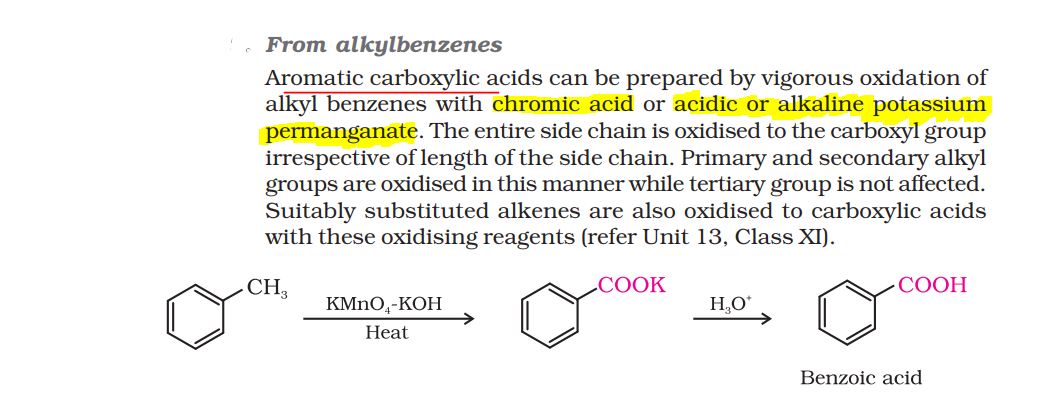
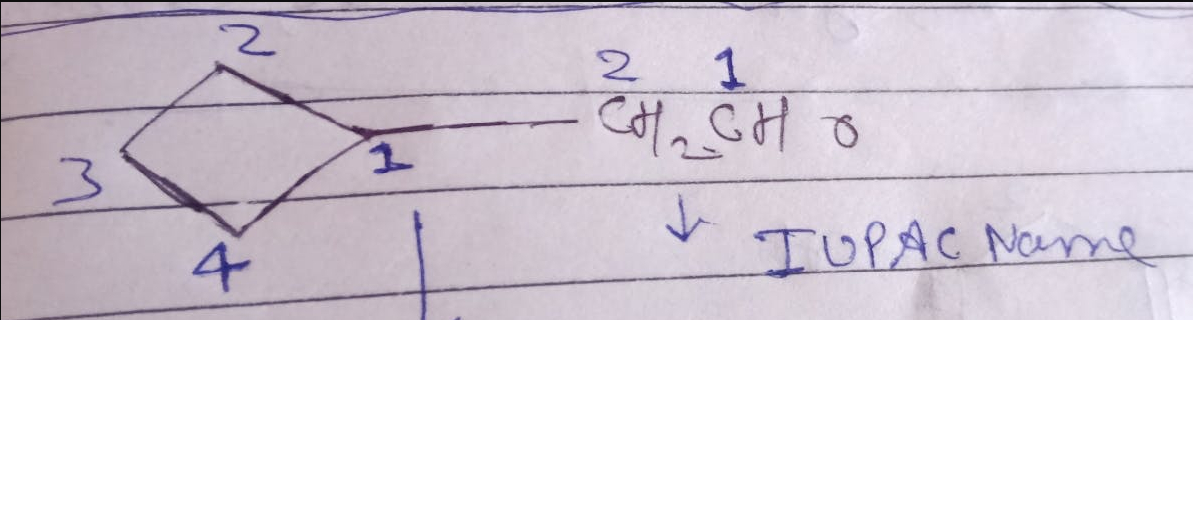
CBSE Class 12-science Answered
How can aldehydes and ketones be prepared by:
i) ozonolysis of alkenes
ii) hydration of alkynes
Asked by padmakrith | 14 Apr, 2013, 11:44: AM
i) ozonolysis of alkenes
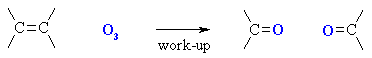
|
||||
REDUCTIVE work-up 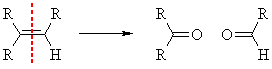 |
OXIDATIVE work-up 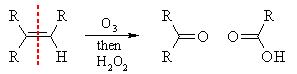 |
|||
ii) hydration of alkynes

Answered by | 15 Apr, 2013, 12:02: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by ukg8612 | 15 Apr, 2024, 07:36: PM
CBSE 12-science - Chemistry
Asked by ajayarchi | 08 Feb, 2024, 03:43: AM
CBSE 12-science - Chemistry
Asked by pallasriramulu9 | 24 Dec, 2023, 06:05: AM
CBSE 12-science - Chemistry
Asked by bsaheliya | 22 Dec, 2023, 09:53: PM
CBSE 12-science - Chemistry
Asked by ygarg8323 | 18 Apr, 2022, 12:47: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 30 Jun, 2021, 04:52: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 28 Jun, 2021, 02:34: PM
CBSE 12-science - Chemistry
Asked by saimerala007 | 22 May, 2021, 02:08: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 31 Dec, 2020, 10:45: AM