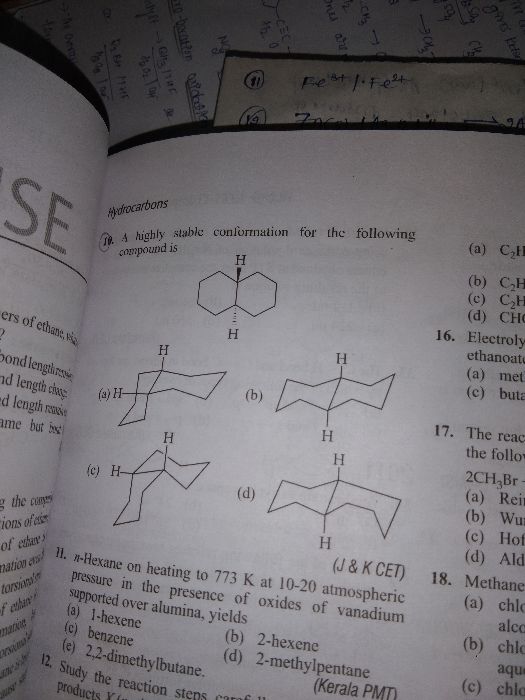
CBSE Class 11-science Answered
Hello ,
Can you please explain the concept of delocalisation of pi electrons ?
Asked by Arun Iyer | 14 Feb, 2011, 10:41: AM
Dear Student
The term 'delocalised' refers to an electron which is not 'attached' to a particular atom. For example, in metals, some of the outer electrons are delocalised and are free to move around the whole metal solid. This is why metals conduct electricity.
Another example of delocalised electrons is in benzene (C6H6), a cyclic molecule composed of a ring of bonded carbons, with one hydrogen attached to each. In benzene, the electrons in the C-C pi-bonds (basically the double bonds) are delocalised the whole molecule. If you look at the location of these pi-bonding electrons, they are found evenly distributed in a ring around the entire molecule. In this case, the delocalisation can be explained by something called 'resonance forms.' Often, molecules with alternating double bonds show delocalised bonding.
However, to truly understand the concept of delocalisation, some basic quantum mechanics must be used, and delocalised bonding in molecules is best explained by molecular orbital theory. In reality, all electrons are somewhat delocalised and are never associated with exactly one bond (in molecules with more than 2 atoms). Some are more localised than others (specific electrons stay mostly with specific bonds), while some are more delocalized (electrons are very free to move about many different bonds in the molecule).
Another example of delocalised electrons is in benzene (C6H6), a cyclic molecule composed of a ring of bonded carbons, with one hydrogen attached to each. In benzene, the electrons in the C-C pi-bonds (basically the double bonds) are delocalised the whole molecule. If you look at the location of these pi-bonding electrons, they are found evenly distributed in a ring around the entire molecule. In this case, the delocalisation can be explained by something called 'resonance forms.' Often, molecules with alternating double bonds show delocalised bonding.
However, to truly understand the concept of delocalisation, some basic quantum mechanics must be used, and delocalised bonding in molecules is best explained by molecular orbital theory. In reality, all electrons are somewhat delocalised and are never associated with exactly one bond (in molecules with more than 2 atoms). Some are more localised than others (specific electrons stay mostly with specific bonds), while some are more delocalized (electrons are very free to move about many different bonds in the molecule).
We hope that clarifies your query.
Regards
Team
Topperlearning
Answered by | 15 Feb, 2011, 10:49: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by kirithshiv | 24 Feb, 2024, 12:12: PM
CBSE 11-science - Chemistry
Asked by shivanij4734 | 16 Dec, 2023, 08:30: PM
CBSE 11-science - Chemistry
Asked by maibamjohnny89 | 15 Jan, 2022, 09:38: PM
CBSE 11-science - Chemistry
Asked by archu312004 | 07 Feb, 2021, 10:21: PM
CBSE 11-science - Chemistry
Asked by dubeyanubhav65 | 18 Jan, 2021, 09:52: PM
CBSE 11-science - Chemistry
Asked by shahsaqlain107 | 30 Nov, 2020, 01:10: PM
CBSE 11-science - Chemistry
Asked by ghastipratiksha | 11 Jul, 2020, 08:14: PM
CBSE 11-science - Chemistry
Asked by guptaserendri | 01 Jul, 2020, 03:58: PM
CBSE 11-science - Chemistry
Asked by Manpreetsingh669933 | 13 Apr, 2020, 01:39: PM
CBSE 11-science - Chemistry
Asked by bittutiwary1234 | 23 Feb, 2020, 12:15: AM