CBSE Class 12-science Answered
Calculate the amount of CaCl2, which must be added to 2 kg water so that the freezing point is depressed by 61 K (Kf of H2O =1.86 K kg mol^-1, Atomic mass of Ca = 40, Cl = 35.5)
Asked by jusu20111 | 26 Jan, 2020, 13:15: PM
Depression in freezing point can be calculated by-
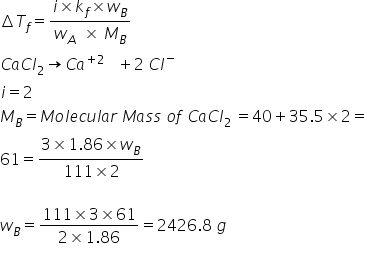
Answered by Ravi | 27 Jan, 2020, 12:12: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by chetanrakshit06 | 05 May, 2024, 14:51: PM
CBSE 12-science - Chemistry
Asked by varinder2149 | 10 Dec, 2023, 20:21: PM
CBSE 12-science - Chemistry
Asked by vekariyaparth61 | 16 May, 2022, 16:33: PM
CBSE 12-science - Chemistry
Asked by goyalpavitarta | 03 May, 2021, 13:27: PM
CBSE 12-science - Chemistry
Asked by goyalpavitarta | 03 May, 2021, 13:25: PM
CBSE 12-science - Chemistry
Asked by goyalpavitarta | 03 May, 2021, 13:20: PM
CBSE 12-science - Chemistry
Asked by sshashu993 | 25 Jul, 2020, 08:02: AM
CBSE 12-science - Chemistry
Asked by anukritisingh8103.bmps | 15 Jul, 2020, 17:52: PM
CBSE 12-science - Chemistry
Asked by sharmasherryal | 25 May, 2020, 09:54: AM