CBSE Class 12-science Answered
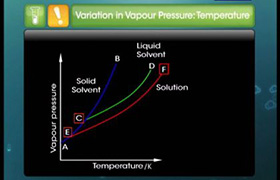
Boiling point of water at 750mm of hg is 99.63°C. how much sucrose is to be added to 500g of water such that it boils at 100°C.???????
Asked by goyalpavitarta | 03 May, 2021, 13:25: PM
Molal elevation constant for water is 0.52 K Kg mol-1.

Answered by Ravi | 03 May, 2021, 16:34: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by chetanrakshit06 | 05 May, 2024, 14:51: PM
CBSE 12-science - Chemistry
Asked by varinder2149 | 10 Dec, 2023, 20:21: PM
CBSE 12-science - Chemistry
Asked by vekariyaparth61 | 16 May, 2022, 16:33: PM
CBSE 12-science - Chemistry
Asked by goyalpavitarta | 03 May, 2021, 13:27: PM
CBSE 12-science - Chemistry
Asked by goyalpavitarta | 03 May, 2021, 13:25: PM
CBSE 12-science - Chemistry
Asked by goyalpavitarta | 03 May, 2021, 13:20: PM
CBSE 12-science - Chemistry
Asked by sshashu993 | 25 Jul, 2020, 08:02: AM
CBSE 12-science - Chemistry
Asked by anukritisingh8103.bmps | 15 Jul, 2020, 17:52: PM
CBSE 12-science - Chemistry
Asked by sharmasherryal | 25 May, 2020, 09:54: AM