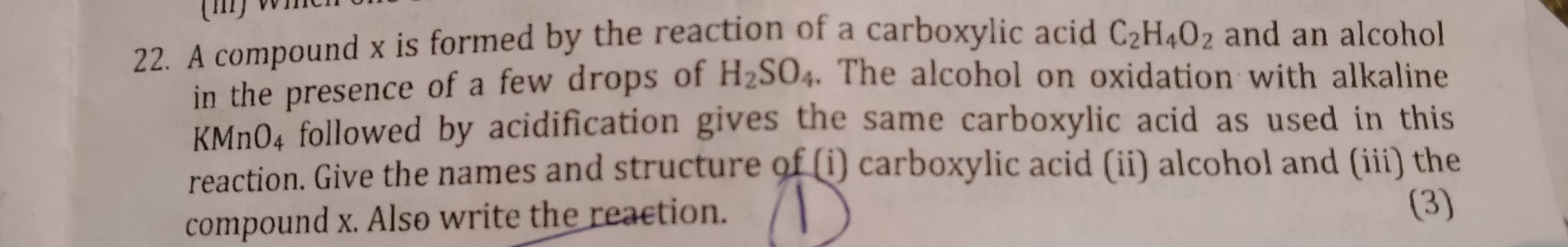CBSE Class 10 Answered
Bacl2 + Na3Po4 - Nacl + Ba3(Po4)2
If 0.5 moles of BAcl2 is mixed with 0.2 moles of Na3Po4 the maximum no.of moles of Ba3(Po4)2that can be formed is
Asked by vedant sharma | 30 Oct, 2014, 04:01: PM
Balanced reaction is:
3BaCl2 + 2Na3PO4 → Ba3(PO4)2 + 6NaCl
3 moles of BaCl2 react with 2 moles of Na3PO4 to give 1 mole of Ba3(PO4)2
The mole ratio between BaCl2 and Na3PO4 is
n(BaCl2):n(Na3PO4)=3:2
The ratio of these compounds in the given mixture is 0.5/0.2 = 5:2 > 3:2.
Hence, the BaCl2 is in excess and the Na3PO4 is the limiting reactant.Now, 2 moles of Na3PO4 give 1 mole of Ba3(PO4)2
So, 0.2 moles of Na3PO4 will give (1/2) x 0.2 = 0.1 mole of Ba3(PO4)2
So, number of moles of Ba3(PO4)2 formed = 0.1
Answered by Arvind Diwale | 31 Oct, 2014, 10:56: AM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by sneh | 27 Mar, 2020, 10:11: AM
CBSE 10 - Chemistry
Asked by pranjaliinamdar2004 | 29 Feb, 2020, 07:20: PM
CBSE 10 - Chemistry
Asked by sweetykhatri99254 | 27 Feb, 2020, 03:40: PM
CBSE 10 - Chemistry
Asked by priyanshiishu | 30 Jan, 2020, 10:39: AM
CBSE 10 - Chemistry
Asked by kamalnayansingh7 | 13 Jan, 2020, 08:35: AM
CBSE 10 - Chemistry
Asked by Deepak | 22 Dec, 2019, 11:20: PM
CBSE 10 - Chemistry
Asked by ritikraghuwanshi6986 | 16 Dec, 2019, 08:42: PM
CBSE 10 - Chemistry
Asked by vedantsagrawal23 | 05 Dec, 2019, 08:34: AM
CBSE 10 - Chemistry
Asked by aryasaxena2003 | 25 Jul, 2019, 05:35: PM
CBSE 10 - Chemistry
Asked by rushabhjain.avv | 21 Mar, 2019, 10:07: PM