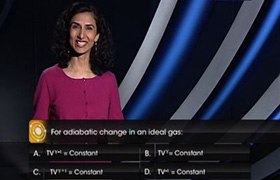
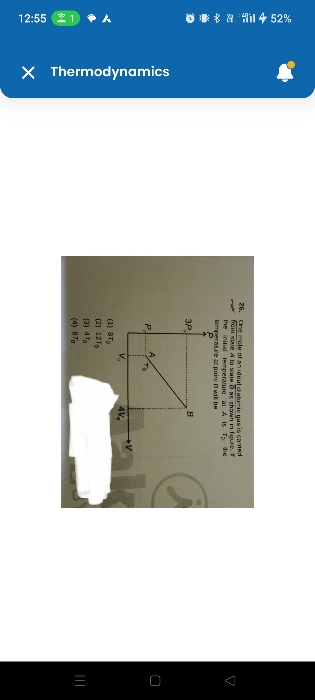
CBSE Class 11-science Answered
(a) Write the isothermal relation between pressure and volume.
(b) Write an expression for work done during isothermal change.
Asked by Topperlearning User | 17 Apr, 2015, 10:09: AM
(a) Pressure x Volume = Constant
or PV = Constant
(b) Work done is
W = μRT ln(V2/V1)
where R is the universal gas constant, μ is the number of moles of gas, T is the absolute temperature, V1 is the initial volume and V2 is the final volume of the gas.
Answered by | 17 Apr, 2015, 12:09: PM
Concept Videos
CBSE 11-science - Physics
Asked by shubham23302007 | 23 Jan, 2024, 22:24: PM
CBSE 11-science - Physics
Asked by s3043632 | 22 Jan, 2023, 18:45: PM
CBSE 11-science - Physics
Asked by govtsecschoolnayaganv051 | 14 Dec, 2018, 19:19: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 17 Apr, 2015, 10:09: AM
CBSE 11-science - Physics
Asked by Topperlearning User | 17 Apr, 2015, 10:11: AM
CBSE 11-science - Physics
Asked by araima2001 | 17 Mar, 2017, 08:12: AM
CBSE 11-science - Physics
Asked by Kusum and Sanjeet | 13 Jul, 2015, 22:02: PM