CBSE Class 10 Answered
A metal carbonate X on treatment with a mineral acid liberates a gas which when passed through aqueous solution of a substance Y gives back X. The substance Y on reaction with the gas obtained at anode during electrolysis of brine gives a compound Z which can decolorise coloured fabrics. Name the compounds X,Y and Z ?
Asked by rushabh123 | 14 Mar, 2019, 10:57: AM
A metal carbonate X on treatment with a mineral acid liberates a gas which when passed through an aqueous solution of a substance Y gives back X.
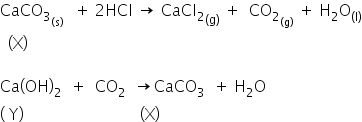
Chlorine gas is obtained at the anode during electrolysis of brine.
The substance Y on reaction with the Cl2 gas obtained at the anode during electrolysis of brine gives a compound Z.
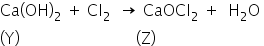
The compound X is CaCO3
The compound y is Ca(OH)2
The compound Z is CaOCl2
Answered by Varsha | 14 Mar, 2019, 03:23: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by p6917110 | 29 Oct, 2023, 09:06: AM
CBSE 10 - Chemistry
Asked by namyabachhal | 16 Oct, 2023, 10:17: PM
CBSE 10 - Chemistry
Asked by chetnasachdeva.sachdeva2006 | 25 Nov, 2021, 05:26: PM
CBSE 10 - Chemistry
Asked by Trisha Gupta | 23 Sep, 2021, 06:56: AM
CBSE 10 - Chemistry
Asked by aryac2904 | 24 Jul, 2021, 08:42: PM
CBSE 10 - Chemistry
Asked by satyendraprajapati6285 | 29 Mar, 2021, 08:40: AM
CBSE 10 - Chemistry
Asked by ShauryaSaxena1011 | 15 Feb, 2021, 08:26: PM
CBSE 10 - Chemistry
Asked by ayushkashyap13052002 | 30 Aug, 2020, 09:10: AM
CBSE 10 - Chemistry
Asked by vandana.bihari.vb | 24 May, 2020, 03:07: PM