CBSE Class 12-science Answered
A hydride of gr 16 element is a gas and acts as a reducing agent when reacted with SO2. It has a smell of rotten eggs and is used in qualitative analysis. It forms a black ppt with Ni2+ in basic medium. It also forms a black ppt with lead acetate solution. Identify the gas and write the equations involved.
Asked by lekhakarthikeyan | 28 Aug, 2018, 04:01: AM
1) A hydride of gr 16 elements is a gas and acts as a reducing agent when reacted with SO2. It has a smell of rotten eggs and is used in qualitative analysis.
There is only one hydride which has rotten eggs smell and is used in the qualitative analysis is H2S.
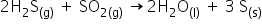
2) It forms a black ppt with Ni2+ in basic medium. It also forms a black ppt with lead acetate solution
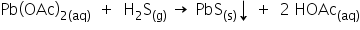
Answered by Ramandeep | 29 Aug, 2018, 11:46: AM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by gurugubellisaivishal2705 | 09 Jul, 2022, 12:16: PM
CBSE 12-science - Chemistry
Asked by cute44464 | 01 Mar, 2021, 13:17: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 26 Feb, 2020, 10:00: AM
CBSE 12-science - Chemistry
Asked by asurve | 10 Sep, 2019, 23:57: PM
CBSE 12-science - Chemistry
Asked by narayanankunjunni | 14 Feb, 2019, 11:16: AM
CBSE 12-science - Chemistry
Asked by lekhakarthikeyan | 28 Aug, 2018, 04:01: AM
CBSE 12-science - Chemistry
The correct order of S—S bond length in following oxyanions is :
(I) S2O42– (II) S2O52– (III) S2O6–2
Asked by Atulcaald | 18 May, 2018, 01:13: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 07 Jun, 2016, 10:50: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 07 Jun, 2016, 10:54: AM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 07 Jun, 2016, 10:54: AM






