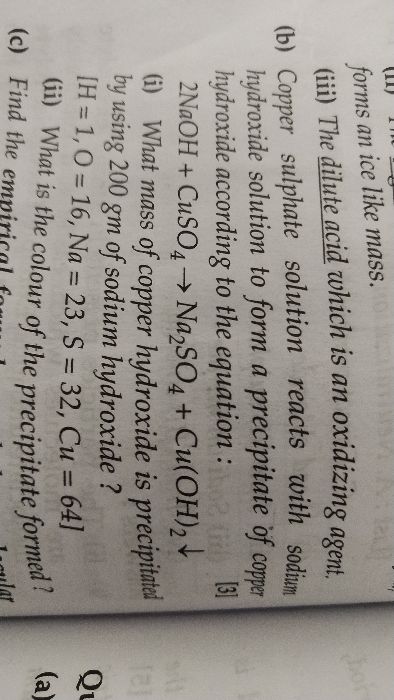ICSE Class 10 Answered
How to know the number of moles of an element in the periodic table?
Asked by reenabhartintpc001 | 12 Mar, 2018, 07:38: PM
Gram atomic mass of an element= 1 mole of element.
We can find the molecular weight from the periodic table.
A mole is a collection of 6.022 × 1023 particles.
A mole is defined as the amount of a substance containing elementary particles like atoms, molecules or ions in 12 gram of carbon-12 (12C).
1 mole of atoms = 6·023 × 1023 atoms = Avogadro’s number
Answered by Varsha | 13 Mar, 2018, 11:41: AM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by jrvedant208 | 05 Feb, 2024, 10:37: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 10 Jul, 2022, 10:13: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 25 Jun, 2022, 10:24: PM
ICSE 10 - Chemistry
Asked by palshivom72 | 14 Jul, 2020, 07:56: PM
ICSE 10 - Chemistry
Asked by jhabijay01 | 27 May, 2020, 12:20: PM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:53: AM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:37: AM
ICSE 10 - Chemistry
Asked by aashimegh | 28 Aug, 2019, 05:25: PM