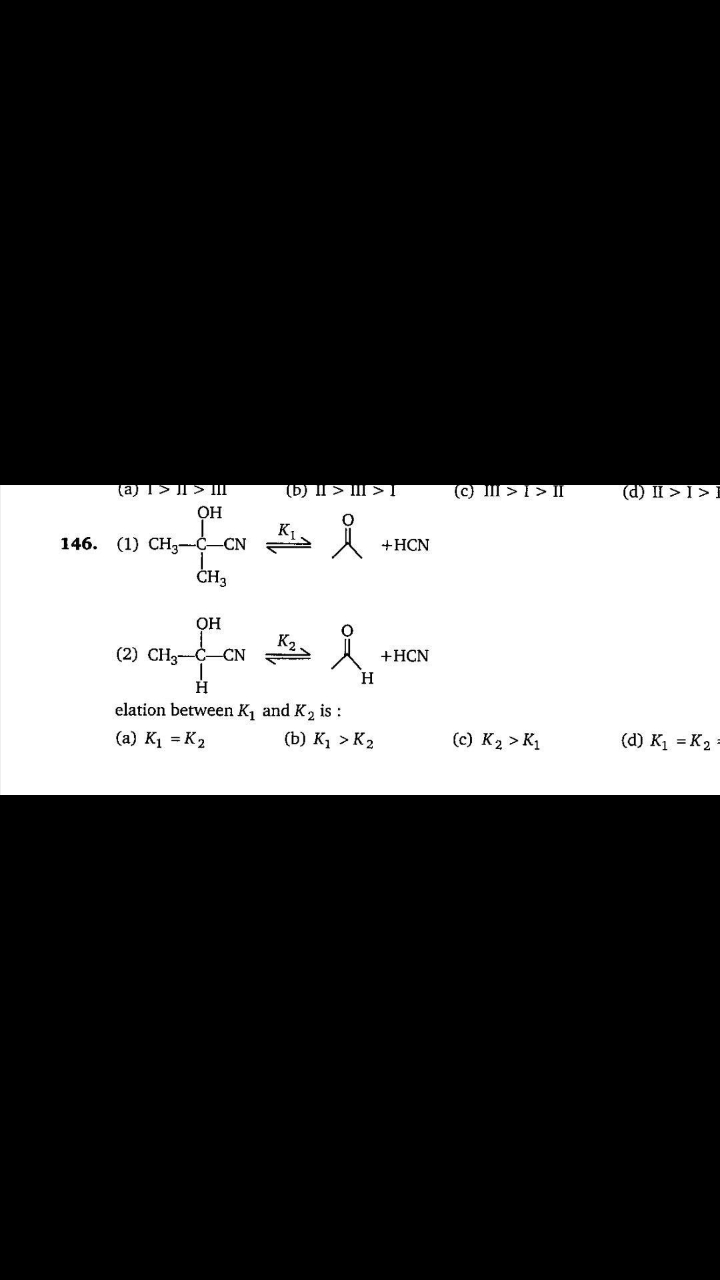
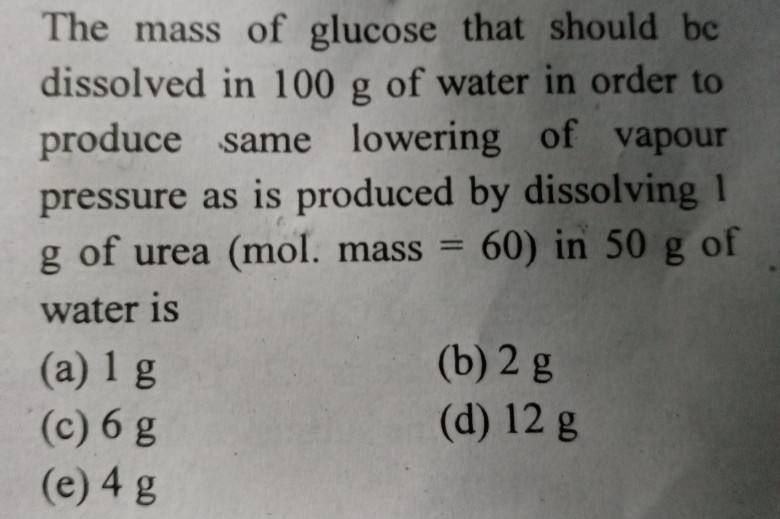NEET Class neet Answered
Solve
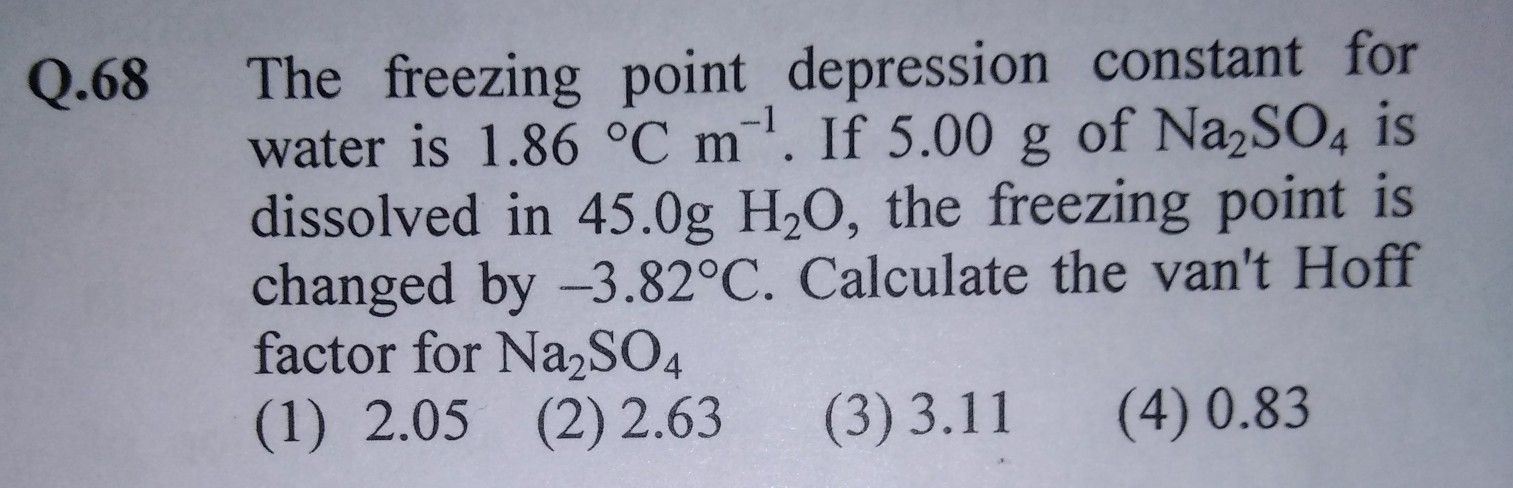
Asked by brijk456 | 03 Sep, 2019, 12:06: AM
Option (2) is correct.
Given:
Kf of water = 1.86 °C m−1
Weight of solute = 5 gm
Weight of solvent = 45 gm
Molar mass of Na2SO4 = 142
ΔTf = −3.82 °C
We know,
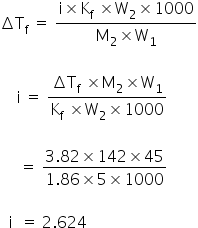
≈ 2.63
van't Hoff factor is 2.63
Answered by Varsha | 03 Sep, 2019, 03:30: PM
Concept Videos
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by ramadevisupriya5678 | 28 Mar, 2024, 02:18: PM
NEET neet - Chemistry
Asked by myindiaisbad | 17 Jun, 2022, 11:17: AM
NEET neet - Chemistry
Asked by bhaveshkaria31 | 30 May, 2022, 09:26: PM
NEET neet - Chemistry
Asked by rautganesh2255 | 01 Jul, 2021, 09:32: AM
NEET neet - Chemistry
Asked by NituBarman192 | 01 Jun, 2021, 10:22: PM
NEET neet - Chemistry
Asked by bhagirathdangi12345 | 12 Feb, 2021, 01:42: PM
NEET neet - Chemistry
Asked by akashmanu09 | 08 Jan, 2021, 10:21: AM
NEET neet - Chemistry
Asked by arnavvidudala20050 | 17 May, 2020, 03:07: PM