CBSE Class 12-science Answered
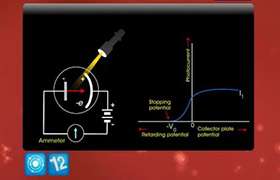
Light of wavelength 500nm falls on a metal whose work function is 1.9 eV . Find (1) the energy of the photon in eV (2) the kinetic energy of the photoelectron emitted and (3) the stopping potential. Given h=6.625x10raise to -34 Js.
Asked by rutujasarangmehta | 15 Mar, 2016, 06:36: PM
λ = 500 nm = 5000 × 10-10
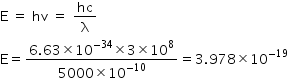
1 eV = 1.6 × 10-19 J
So, 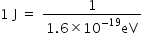
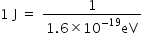
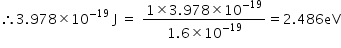
Hence, the energy of the photon is 2.486eV.
E = KE + hv0
'E' is the energy of incident photon, KE is the kinetic energy of emitted photon, hv0 is the work function or minimum energy required.
Work function is 1.9eV.
KE = E - hv0
= 2.486 - 1.9 = 0.586eV
Thus the kinetic energy of the photoelectron emitted is 0.586eV.
Answered by Faiza Lambe | 16 Mar, 2016, 01:25: PM
Concept Videos
CBSE 12-science - Physics
Asked by mishrigupta19319 | 08 Apr, 2024, 06:28: PM
CBSE 12-science - Physics
Asked by mishrigupta19319 | 07 Apr, 2024, 11:23: AM
CBSE 12-science - Physics
Asked by shivakumarshreyas24 | 01 Mar, 2020, 08:12: AM
CBSE 12-science - Physics
Asked by khushimassey437 | 31 May, 2019, 08:41: AM
CBSE 12-science - Physics
Asked by manasvijha | 19 Mar, 2019, 07:17: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM