CBSE Class 11-science Answered
Calculate the no. Of atoms
A) 5.6 L of NH3
B) 4.4g of Co2
C) 52u of He atomic weight =4u)
Asked by rajdhan.yadav187350 | 12 Nov, 2018, 02:05: PM
A) 5.6 litre of NH3

B) 4.4 g of CO2

C) 52 u of He
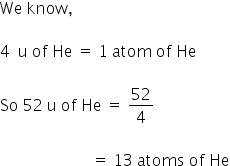
5.6 litre of NH3 contains 6×1023 atoms of NH3
4.4 g of CO2 contains 1.8 ×1023 atoms of CO2
52 u of He contains 13 atoms of He.
Answered by Varsha | 13 Nov, 2018, 11:25: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by manikandanragul1 | 11 Apr, 2024, 09:02: AM
CBSE 11-science - Chemistry
Asked by nikhithaguguloth14 | 29 Mar, 2024, 08:15: PM
CBSE 11-science - Chemistry
Asked by sumedhasingh238 | 27 Mar, 2024, 11:04: PM
CBSE 11-science - Chemistry
Asked by avijotsingh946431 | 22 Feb, 2024, 05:36: PM
CBSE 11-science - Chemistry
Asked by gurmelsinghray | 21 Feb, 2024, 08:43: AM
CBSE 11-science - Chemistry
Asked by bablipanwar893 | 01 Jul, 2023, 12:25: PM
CBSE 11-science - Chemistry
Asked by saijagdale9 | 19 Jun, 2023, 02:34: PM
CBSE 11-science - Chemistry
Asked by kdimple765 | 17 Jul, 2022, 01:24: PM
CBSE 11-science - Chemistry
Asked by alfirozislam900 | 03 Jul, 2022, 01:24: PM
CBSE 11-science - Chemistry
Asked by alfirozislam900 | 03 Jul, 2022, 01:23: PM




