CBSE Class 12-science - Rate of Chemical Reaction Videos
Calculate rate of reactions
Define and calculate rate, average & instantaneous rate. Express rate of reaction in terms of stoichiometric coefficients.
More videos from this chapter
View All- Explain SN2 Mechanism
- effect of cone temperature of the rate of b/w sodium thiosulphur and hydrochloric acid.
- distinction between rate of reaction and rate constant
- Of 50% of reaction gets completed in 16 minutes what fraction of reaction would occur in 32 minutes
-
Please solve question 25

- Write integrated rate expression for the first order reaction. Also, findHalf life for this expression.
-
the given questions

- rate equation
-
pls solve
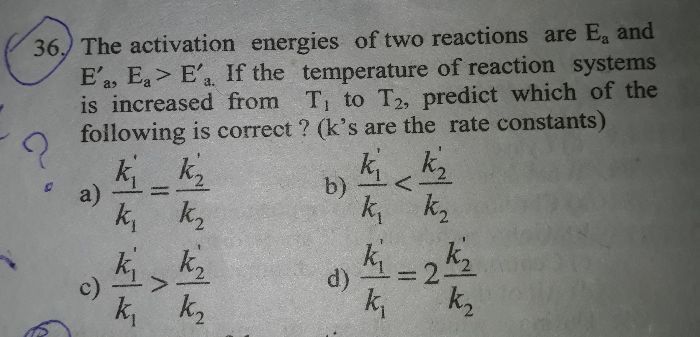
- In the reaction A + 2B ??2C + D. If the concentration of A is increased four times and B is decreased to half of its initial concentration then the rate becomes






