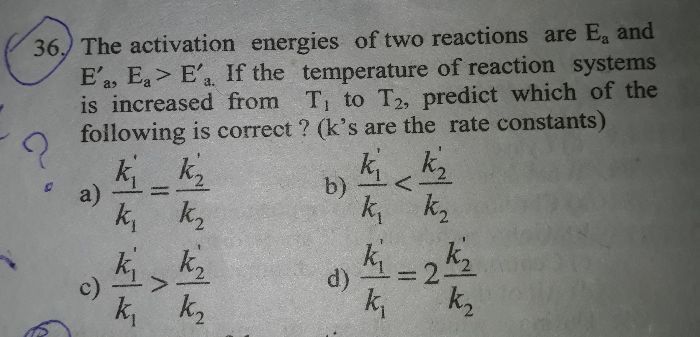CBSE Class 12-science Answered
In the reaction A + 2B ??2C + D. If the concentration of A is increased four times and B is decreased to half of its initial concentration then the rate becomes
Asked by leelakrishnapallapotu143 | 29 Mar, 2020, 19:27: PM
Rate of reaction can be calculated by
R=[A]x [B]y
R1=k[A]1 [B]2
R2 =k[4A][B/2]2
R2 /R1 =1
Rate will be unchenged
Answered by Ravi | 31 Mar, 2020, 19:21: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by desaianant541 | 15 May, 2024, 21:05: PM
CBSE 12-science - Chemistry
Asked by bhadauriyax | 30 Nov, 2023, 18:23: PM
CBSE 12-science - Chemistry
Asked by rahulbiswal946 | 08 Nov, 2023, 19:01: PM
CBSE 12-science - Chemistry
Asked by arshbhatia0809 | 22 Jul, 2021, 21:47: PM
CBSE 12-science - Chemistry
Asked by Surendersingh0493 | 18 Oct, 2020, 14:05: PM
CBSE 12-science - Chemistry
Asked by khandarev3580 | 10 Oct, 2020, 10:54: AM
CBSE 12-science - Chemistry
Asked by dr.akanksha0411 | 07 Aug, 2020, 11:56: AM
CBSE 12-science - Chemistry
Asked by amritha2960 | 13 May, 2020, 08:26: AM
CBSE 12-science - Chemistry
Asked by sulaikhasulu393 | 02 May, 2020, 09:20: AM
CBSE 12-science - Chemistry
Asked by leelakrishnapallapotu143 | 29 Mar, 2020, 19:27: PM