CBSE Class 12-science Answered
Sir,
can you please tell me that how to find the order of reactants while calculating order of reaction. Suppose a reaction
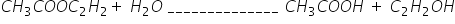 in this reaction how do we know the order of reactants????????????????
in this reaction how do we know the order of reactants????????????????
Asked by PRIYANKAJ YADAV | 17 Feb, 2015, 08:28: AM
Dear jepankajyadav123@rediffmail.com
Thanks for asking us a question in Ask the Expert section of TopperLearning.com.
The reaction rate law expression relates the rate of a reaction to the concentrations of the reactants.
aA + bB → cC + dD
Rate = k [A]x [B]y
x and y are the reactant orders determined from experiment. x and y are NOT the stoichiometric coefficients. The rate law is always determined experimentally. There are several ways to determine the rate law for a particular reaction like ‘Method of Initial Rates’ and ‘Isolation Method’.
In the hydrolysis of ethyl acetate water is added in excess so the rate is dependant only on the concentration of ethyl acetate so it is a first order reaction w.r.t to ethyla acetate.
Regards
Topperlearning Team.
Answered by Arvind Diwale | 17 Feb, 2015, 01:09: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by bhadauriyax | 30 Nov, 2023, 06:23: PM
CBSE 12-science - Chemistry
Asked by rahulbiswal946 | 08 Nov, 2023, 07:01: PM
CBSE 12-science - Chemistry
Asked by mishraridhi2020 | 23 Jun, 2022, 09:16: AM
CBSE 12-science - Chemistry
Asked by cjam41665 | 10 Oct, 2021, 12:56: AM
CBSE 12-science - Chemistry
Asked by arshbhatia0809 | 22 Jul, 2021, 09:47: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 14 Jan, 2021, 12:40: PM
CBSE 12-science - Chemistry
Asked by Surendersingh0493 | 18 Oct, 2020, 02:05: PM
CBSE 12-science - Chemistry
Asked by khandarev3580 | 10 Oct, 2020, 10:54: AM
CBSE 12-science - Chemistry
Asked by rchaitra1204 | 07 Sep, 2020, 09:43: AM
CBSE 12-science - Chemistry
Asked by dr.akanksha0411 | 07 Aug, 2020, 11:56: AM










