ICSE Class 10 Answered
Q.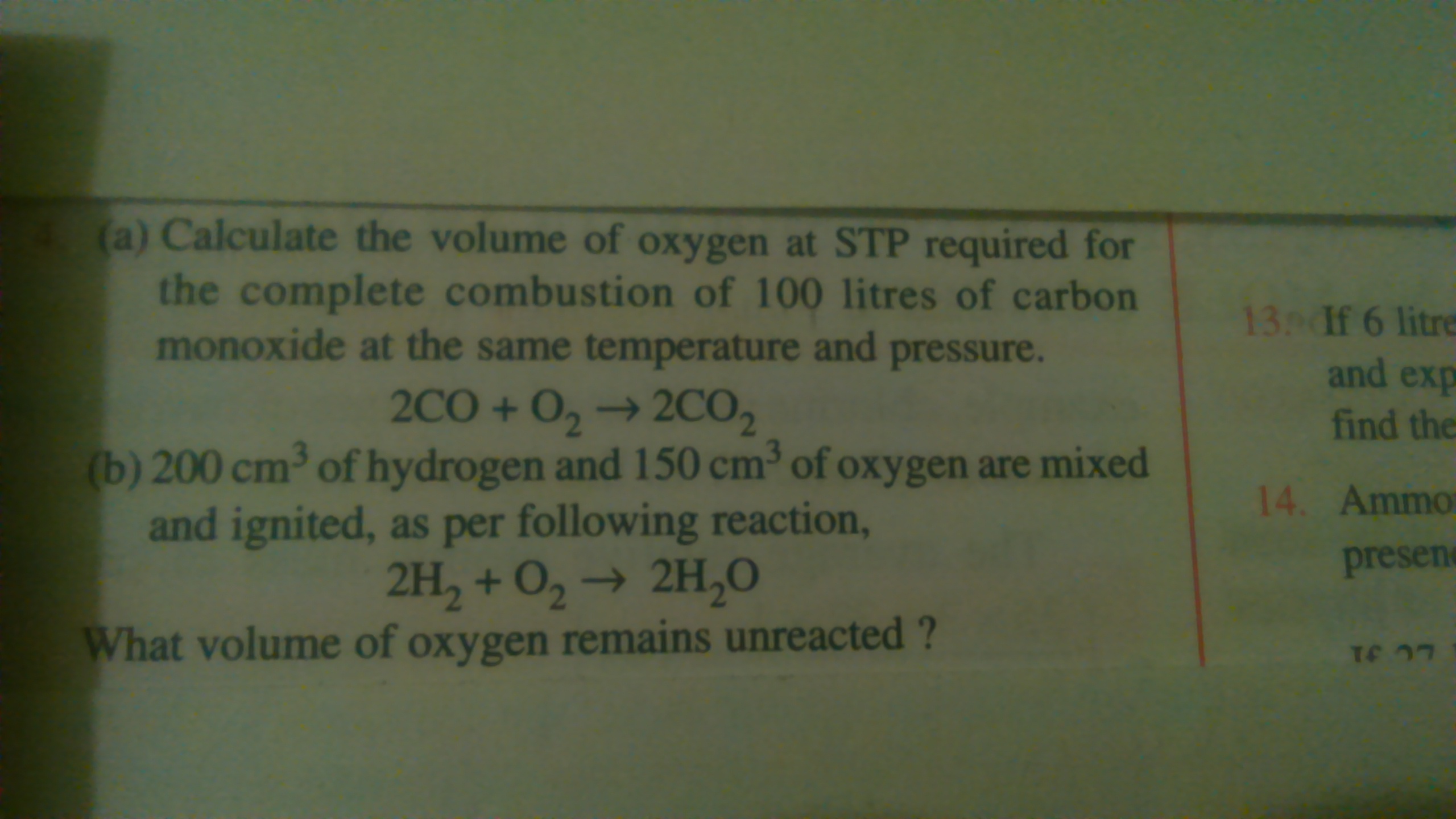
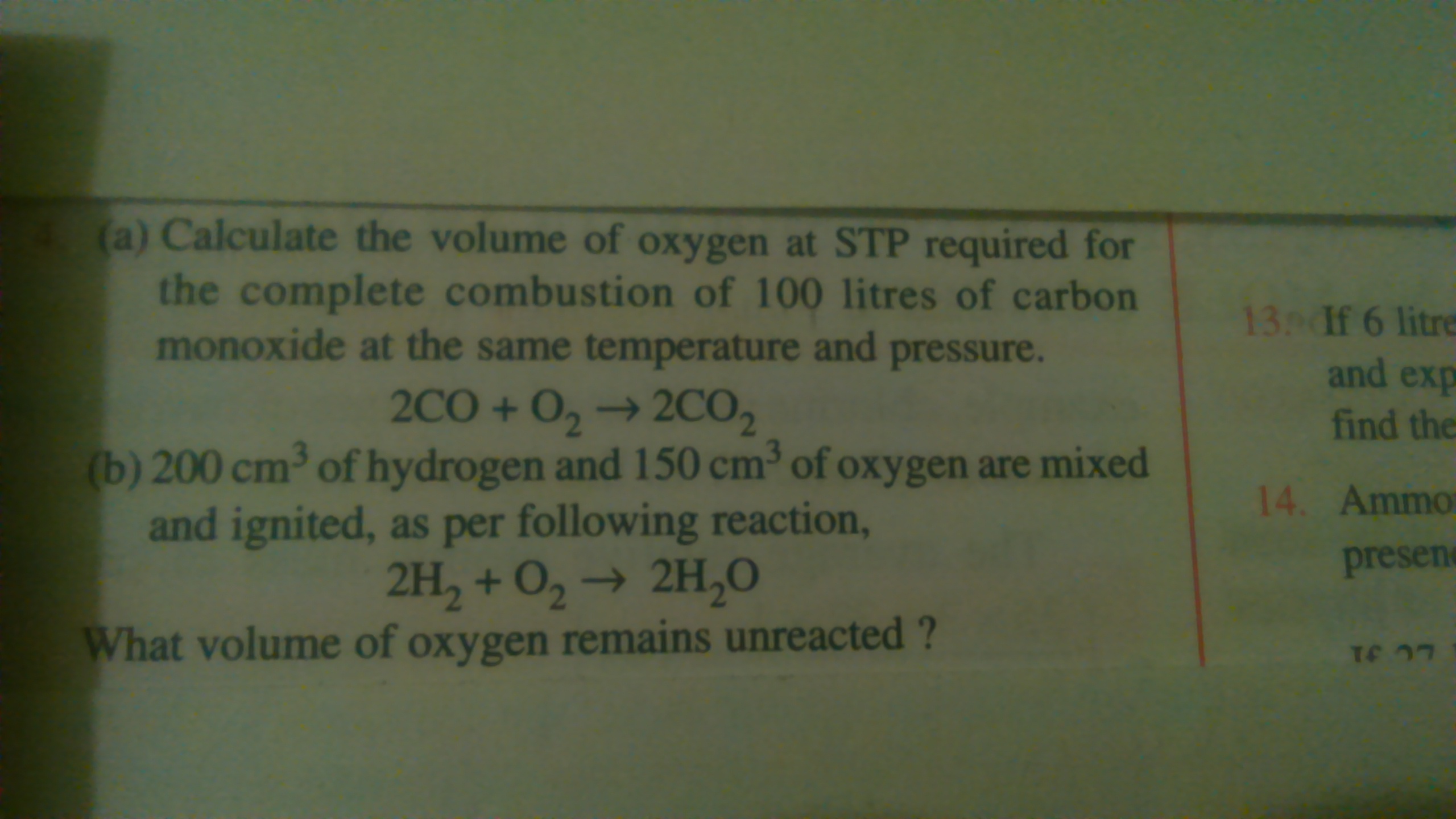
Asked by Invest123736 | 23 May, 2016, 21:54: PM
(a)
2CO + O2 → 2CO2
2 V 1 V 2 V
2 V of CO requires = 1V of O2
so, 100 litres of CO requires = 50 litre of O2
(b)
2H2 + O2 → 2H2O
2 V 1V 2V
From the equation, 2V of hydrogen reacts with 1V of oxygen
so 200cm3 of Hydrogen reacts with = 200/2= 100 cm3
Hence, the unreacted oxygen is 150 - 100 = 50cm3 of oxygen.
Answered by Vaibhav Chavan | 24 May, 2016, 10:48: AM
Concept Videos
ICSE 10 - Chemistry
Asked by ruchisharmatbn | 05 Mar, 2024, 18:40: PM
ICSE 10 - Chemistry
Asked by kundus458 | 07 Feb, 2024, 08:55: AM
ICSE 10 - Chemistry
Asked by matloobser | 07 Sep, 2023, 09:36: AM
ICSE 10 - Chemistry
Asked by dafk04.dp | 06 May, 2021, 18:22: PM
ICSE 10 - Chemistry
Asked by amit.clw4 | 15 Mar, 2021, 07:27: AM
ICSE 10 - Chemistry
Asked by amit.clw4 | 14 Mar, 2021, 08:12: AM
ICSE 10 - Chemistry
Asked by ravi.solaabhi | 17 Oct, 2020, 10:11: AM
ICSE 10 - Chemistry
Asked by payalagrawal1724 | 28 Jun, 2020, 19:22: PM
ICSE 10 - Chemistry
Asked by vijay.prag | 29 Dec, 2019, 20:07: PM
ICSE 10 - Chemistry
Asked by Dsangayy | 08 May, 2019, 19:11: PM






