NEET Class neet Answered
In which of the following transition the wavelength be minimum ?
a)n=5 to n=4
b)n=4 to n=3
c)n=3 to n=2
d)n=2 to n=1
Asked by akshadevdm2020 | 22 May, 2021, 15:43: PM
Wavelength 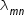 of spectral line that arises due to transition from orbital quantum number m to orbital quantum number n is given as
of spectral line that arises due to transition from orbital quantum number m to orbital quantum number n is given as
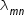 of spectral line that arises due to transition from orbital quantum number m to orbital quantum number n is given as
of spectral line that arises due to transition from orbital quantum number m to orbital quantum number n is given as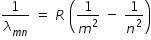
where R is Rydberg constant .
Reciprocal of wavelength for all the transitions are tabulated below
| Transition | Eqn. for (1/λ) | 1/λ |
| n=5 to n=4 |
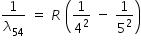 |
R × 0.0225 |
| n = 4 to n = 3 |
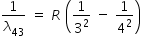 |
R × 0.0486 |
| n = 3 to n = 2 |
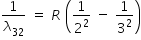 |
R × 0.1389 |
| n = 2 to n = 1 |
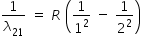 |
R × 0.5 |
From above table , it can be seen that for the transition from n=2 to n=1 , Reciprocal of wavelength is maximum.
Hence wavelength is minimum for transition from n=2 to n=1
Answered by Thiyagarajan K | 22 May, 2021, 16:56: PM
Concept Videos
NEET neet - Physics
Asked by roshanrocky334 | 13 Jan, 2024, 11:52: AM
NEET neet - Physics
Asked by adititiwari601 | 13 Jun, 2022, 07:44: AM
NEET neet - Physics
Asked by begfatima123 | 06 May, 2022, 23:37: PM
NEET neet - Physics
Asked by jhajuhi19 | 30 Aug, 2021, 20:02: PM
NEET neet - Physics
Asked by akshadevdm2020 | 22 May, 2021, 15:43: PM
NEET neet - Physics
Asked by akdwadasi1111 | 20 Apr, 2021, 12:19: PM
NEET neet - Physics
Asked by Prashant DIGHE | 10 Apr, 2020, 21:21: PM