ICSE Class 10 Answered
In lone pair effect of oxygen atom
When H+combines with H2O why the hydronium ion gets positive charge
Asked by lovemaan5500 | 22 Nov, 2017, 06:38: AM
oxygen in the water molecule has the lone pair of electrons, oxygen donates its lone pair to H+ ions which has lack of electrons and formed positively charged hydronium ion
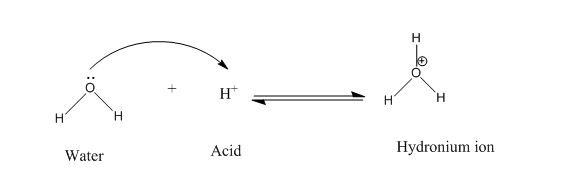
Answered by Ramandeep | 22 Nov, 2017, 10:42: AM
Concept Videos
ICSE 10 - Chemistry
Asked by vijayvijay09644 | 06 Mar, 2024, 10:37: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 18 Jul, 2022, 10:39: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 18 Jul, 2022, 10:33: PM
ICSE 10 - Chemistry
Asked by manasa | 10 Sep, 2021, 06:32: PM
ICSE 10 - Chemistry
Asked by waliaman704 | 29 Jun, 2021, 11:58: AM
ICSE 10 - Chemistry
Asked by manbeersinghahhps | 19 May, 2021, 07:12: PM
ICSE 10 - Chemistry
Asked by aras89009 | 09 May, 2021, 02:23: PM
ICSE 10 - Chemistry
Asked by sudhanshutiwari9889 | 29 Sep, 2020, 10:42: PM
ICSE 10 - Chemistry
Asked by adityagogineni15.10spicertl | 17 Jun, 2020, 07:21: PM





