CBSE Class 11-science Answered
For a reaction,
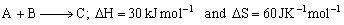 Determine the temperature above which reaction will be spontaneous.
Determine the temperature above which reaction will be spontaneous.
Asked by Topperlearning User | 14 Aug, 2014, 04:53: PM
![]()
Answered by | 14 Aug, 2014, 06:53: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by mankdubey670 | 06 Jun, 2022, 01:27: PM
CBSE 11-science - Chemistry
Asked by gganga | 10 Apr, 2018, 06:02: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 04:44: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 05:46: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 04:51: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 04:53: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 14 Aug, 2014, 05:00: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 05:46: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 23 Sep, 2014, 03:28: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 23 Sep, 2014, 03:39: PM




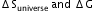 ?
?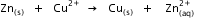 Given that
Given that 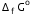 for Cu2+(aq) and Zn2+(aq) as 65 kJ mol-1 and -147.2 kJ mol-1 respectively.
for Cu2+(aq) and Zn2+(aq) as 65 kJ mol-1 and -147.2 kJ mol-1 respectively.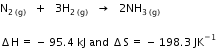 Calculate the temperature at which Gibbs energy change ΔG is equal to zero. Predict the nature of the reaction at this temperature and above it.
Calculate the temperature at which Gibbs energy change ΔG is equal to zero. Predict the nature of the reaction at this temperature and above it.