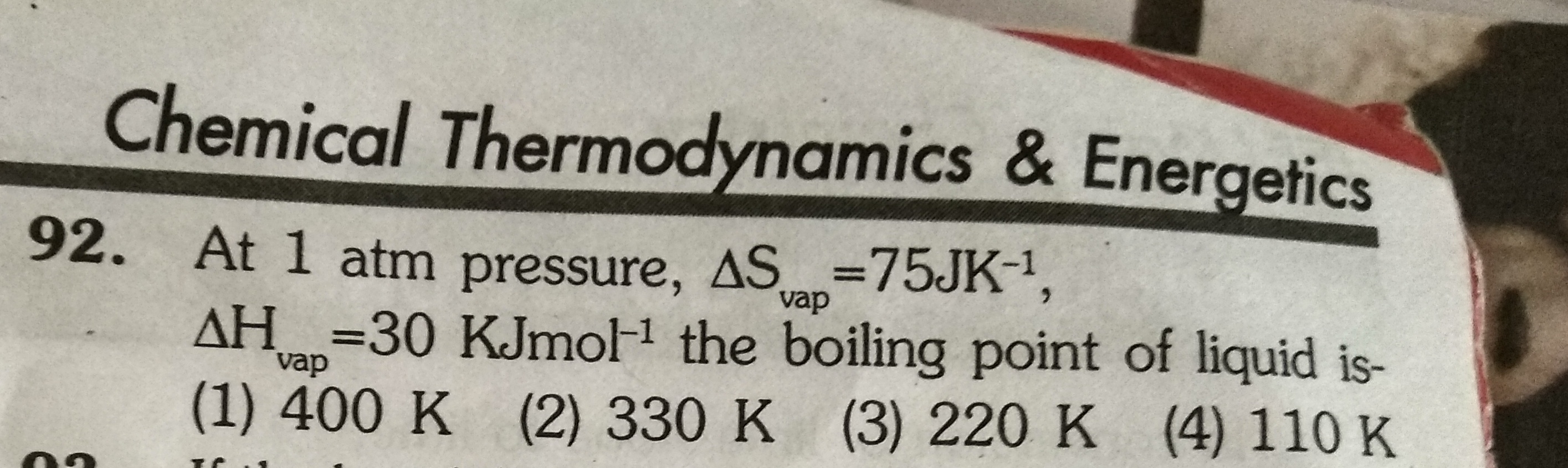JEE Class main Answered
Detailed answers please
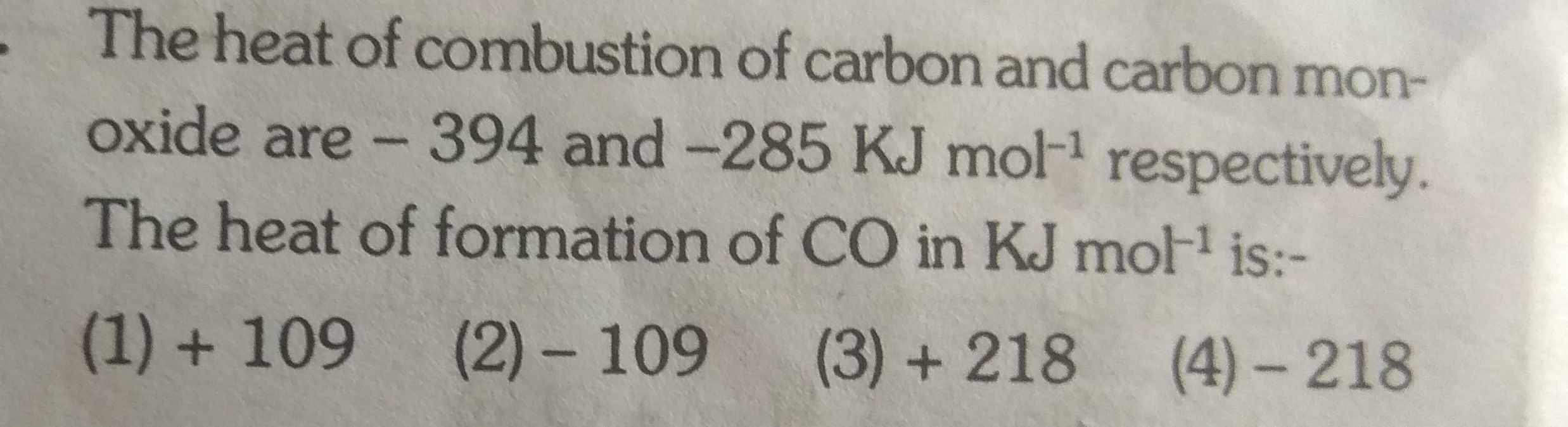
Asked by g_archanasharma | 20 Feb, 2019, 17:23: PM
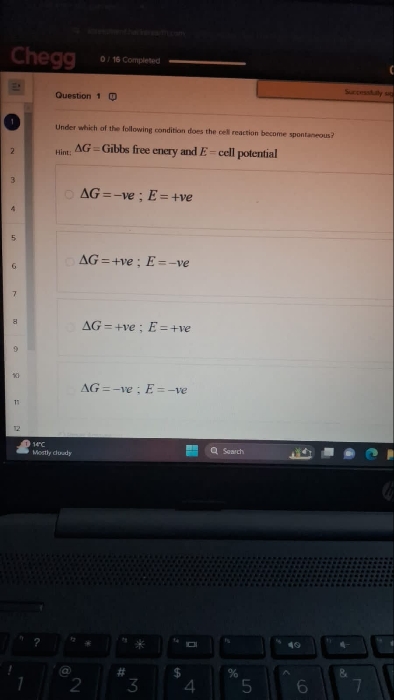
Option (2) is correct.
Combustion of carbon:
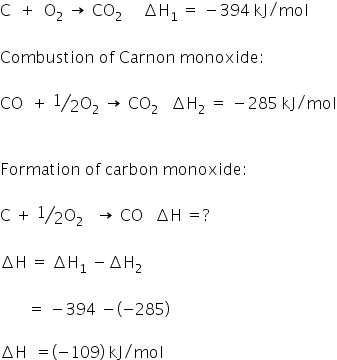
The heat of formation of CO is -109 kJ/mol.
Answered by Varsha | 21 Feb, 2019, 11:16: AM
JEE main - Chemistry
Asked by ashutosharnold1998 | 03 Nov, 2019, 20:22: PM
JEE main - Chemistry
Asked by ashutosharnold1998 | 31 Oct, 2019, 19:16: PM
JEE main - Chemistry
Asked by ashutosharnold1998 | 10 Aug, 2019, 00:10: AM
JEE main - Chemistry
Asked by Ranjeetgupta26068 | 19 May, 2019, 22:14: PM
JEE main - Chemistry
Asked by g_archanasharma | 20 Feb, 2019, 17:23: PM
JEE main - Chemistry
Asked by g_archanasharma | 08 Feb, 2019, 17:48: PM