ICSE Class 8 Answered
An empty RD bottle has a mass of 25.25 g. When filled with water, the bottle has a mass of 55.75 g. and 45.50 g when filled with alcohol. Find the volume of bottle and the RD of alcohol. Also find the density of alcohol.
Asked by sarojpullak | 17 Apr, 2020, 17:35: PM
Mass of empty bottle, mb = 25.25 g
When bottle is filled with water, the mass of bottle is, mbw = 55.75 g
Thus, mass of water, mw = 55.75 - 25.25 g = 30.5 g
When filled with alcohol, mass of the bottle is, mba = 45.50 g
Thus, mass of alcohol, ma = 45.50 - 25.25 = 20.25 g
Volume of bottle = volume occupied by water
We know,
density = mass/ volume
density of water = 1 g/cm3
mass of water = 30.5 g
Thus, volume of water, vw = mw / 1 = 30.5 cm3
Hence,
volume of bottle = 30.5 cm3
volume occupied by water = voulme occupied by alcohol = 30.5 cm3
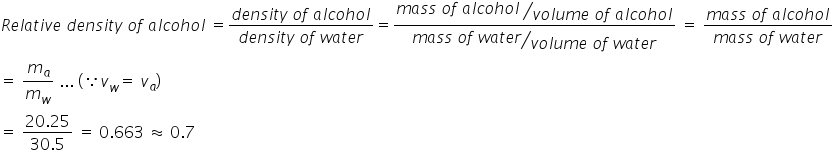
density of alcohol = mass of alcohol/ volume of alcohol = 20.25/30.5 = 0.66 g/cm3 = 0.7 g/cm3
Answered by Shiwani Sawant | 17 Apr, 2020, 19:22: PM
Concept Videos
ICSE 8 - Physics
Asked by authpurnationalmodel88 | 06 Jun, 2024, 15:57: PM
ICSE 8 - Physics
Asked by enakshipal07 | 23 May, 2023, 20:47: PM
ICSE 8 - Physics
Asked by samarthshukla60.8spicertl | 12 May, 2020, 11:15: AM
ICSE 8 - Physics
Asked by sangitshane7758.8sdatl | 05 May, 2020, 09:12: AM
ICSE 8 - Physics
Asked by mandalnikhil0740.8sdatl | 03 May, 2020, 10:44: AM
ICSE 8 - Physics
Asked by yadavvishal222303 | 24 Apr, 2020, 18:59: PM
ICSE 8 - Physics
Asked by sarojpullak | 17 Apr, 2020, 17:35: PM
ICSE 8 - Physics
Asked by rainikita525 | 16 Apr, 2020, 12:16: PM




