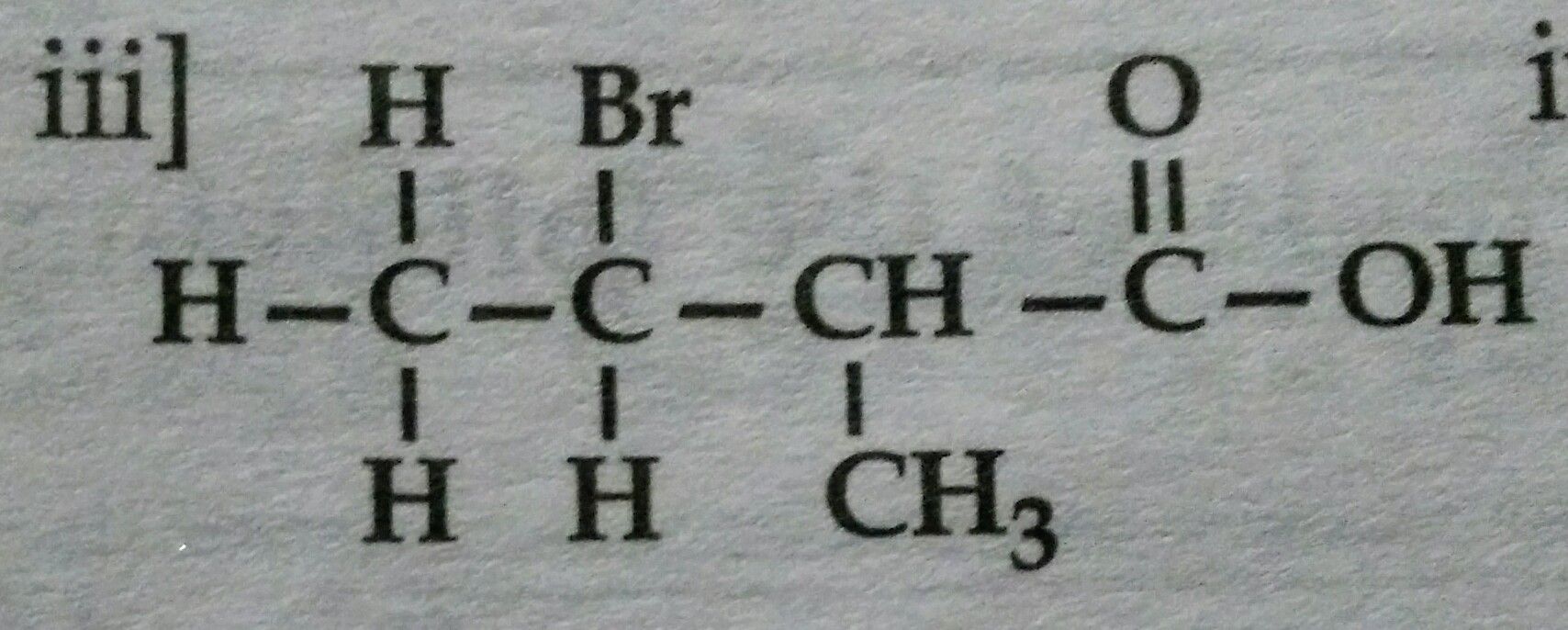ICSE Class 10 Answered
Acetic acid has 4 hydrogen atoms in its molecule but is a monobasic acid.Why?
Asked by swatidutta448 | 21 Jan, 2020, 14:02: PM
Acetic acid has 4 hydrogens. It donates only one H, which is attached to Oxygen, Other 3 H are attached to Carbon, there is not much difference iof electronegativity in C and H in acetic acid so these 3 H can not be released.
So basicity of Acetic acid = 1.
Answered by Ravi | 21 Jan, 2020, 17:50: PM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by singhasees587 | 18 May, 2020, 12:22: PM
ICSE 10 - Chemistry
Asked by mehalsingh | 15 Mar, 2020, 17:34: PM
ICSE 10 - Chemistry
Asked by nusratirfankhan311 | 12 Mar, 2020, 20:49: PM
ICSE 10 - Chemistry
Asked by swatidutta448 | 21 Jan, 2020, 14:02: PM
ICSE 10 - Chemistry
Asked by flaviasaldanha258 | 19 Mar, 2019, 21:05: PM
ICSE 10 - Chemistry
Asked by swayamdesai54689 | 12 Mar, 2019, 18:52: PM
ICSE 10 - Chemistry
Asked by marc21coc | 03 Feb, 2019, 11:34: AM
ICSE 10 - Chemistry
Asked by malapandey8278 | 02 Feb, 2019, 08:54: AM
ICSE 10 - Chemistry
Asked by shamim.ayanahmad | 15 Mar, 2018, 13:55: PM
ICSE 10 - Chemistry
Asked by lovemaan5500 | 18 Jan, 2018, 15:33: PM