CBSE Class 11-science Answered
A school has four storied building. Some students go by staircase whereas some go by the elevator. It is observed that students going by steps feel fresher throughout the day as compared to those who go by the elevator. What values are expressed in the above paragraph
Comment on thermodynamic nature of the work done by two students
Which law application here and how?
Asked by anura_132001 | 22 Oct, 2017, 03:40: PM
Human body obeys the laws of thermodynamics. When we exersise, some of the food energy gets converted into muscle work, but most of energy is converted into thermal energy.
In first case, students are in open system they go by staircase, they start to feel warm & will starts sweating. This is the process their body uses to cool itself means their body maintain equlibrium. Then heat from their body transfer to surrounding, here in this case both 1st and 2nd law of thermodynamic is in action which is no heat is lost, it merely transferred and approches equlibrium with maximum entropy
In case of elevator they are in closed system, their body will undrgo three diffrent equlibrium i.e. before entering elevator, inside the elevator and after coming out of elevator. here 1st and 2nd law of thermodynamics are applicable.
Answered by Ramandeep | 24 Oct, 2017, 06:47: PM
CBSE 11-science - Chemistry
Asked by rukayabatool395 | 24 Mar, 2024, 07:48: PM
CBSE 11-science - Chemistry
Asked by rama26516 | 12 Mar, 2022, 01:49: PM
CBSE 11-science - Chemistry
Asked by mossewalasindhu | 20 Nov, 2021, 02:16: PM
CBSE 11-science - Chemistry
Asked by chandankumargochhayat6 | 19 May, 2021, 08:48: AM
CBSE 11-science - Chemistry
Asked by saimahima2005 | 16 Jul, 2020, 06:28: PM
CBSE 11-science - Chemistry
Asked by Leenatripathi20 | 10 Feb, 2020, 07:12: PM
CBSE 11-science - Chemistry
Asked by ABHILASHA | 26 Aug, 2019, 11:41: PM
CBSE 11-science - Chemistry
Asked by hritikch37 | 16 Dec, 2018, 10:09: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 02:53: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 13 Aug, 2014, 11:18: AM

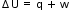 .
.