JEE Class main Answered
Solve

Asked by sarveshvibrantacademy | 12 May, 2019, 01:47: PM
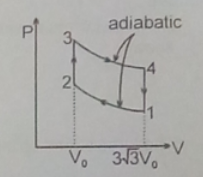
(A) internal energy decreases in the path 3-4
True.
By first law of thermodynamics, dq = du + dw ....................(1)
where dq is the heat energy input , du is change in internal energy and dw is the workdone by the gas.
for adiabatic process, dq = 0, workdone by the gas dw= ∫pdv is positive,
hence du = -dw or internal energy is decreasing.
(B) no work is done during the path 2-3, because volume is constant and change in volume dv = 0
(C) during the path 4-1, volume is constnat.
For ideal gas when volume is constant pressure P is directly proportional to Temperature T.
Since pressure decreasing in the path 4-1, Temperature also devreasing.
(D) during the path 1-2 , workdone dw= ∫pdv , change of volume is negative.
Hence workdone by the gas is negative. Hence work is done on the gas
Answered by Thiyagarajan K | 12 May, 2019, 03:26: PM
Application Videos
JEE main - Physics
Asked by sandhyapallapu22 | 03 May, 2024, 04:32: PM
JEE main - Physics
Asked by gamingbadboy085 | 01 May, 2024, 06:28: PM
JEE main - Physics
Asked by bhargavreddynellaballi | 30 Apr, 2024, 08:18: AM
JEE main - Physics
Asked by ravichandraravula66 | 29 Apr, 2024, 11:20: AM
JEE main - Physics
Asked by kmani310507 | 28 Apr, 2024, 04:38: PM
JEE main - Physics
Asked by arivaryakashyap | 23 Apr, 2024, 10:40: AM
JEE main - Physics
Asked by ratnadeep.dmr003 | 21 Apr, 2024, 11:06: PM
JEE main - Physics
Asked by ksahu8511 | 19 Apr, 2024, 11:55: AM
JEE main - Physics
Asked by mohammedimroz | 13 Apr, 2024, 09:48: PM
JEE main - Physics
Asked by medhamahesh007 | 02 Apr, 2024, 11:11: AM