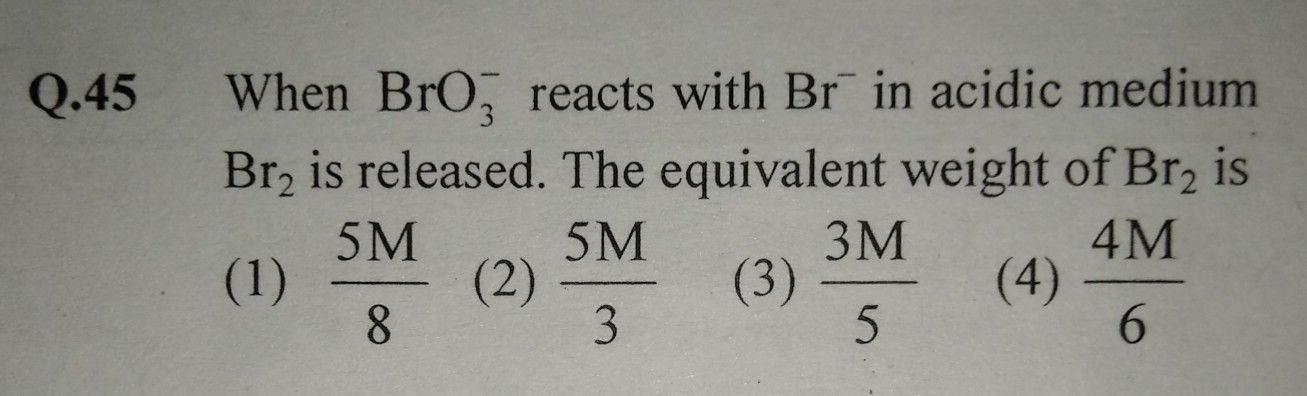NEET Class neet Answered
Solve it sir ji
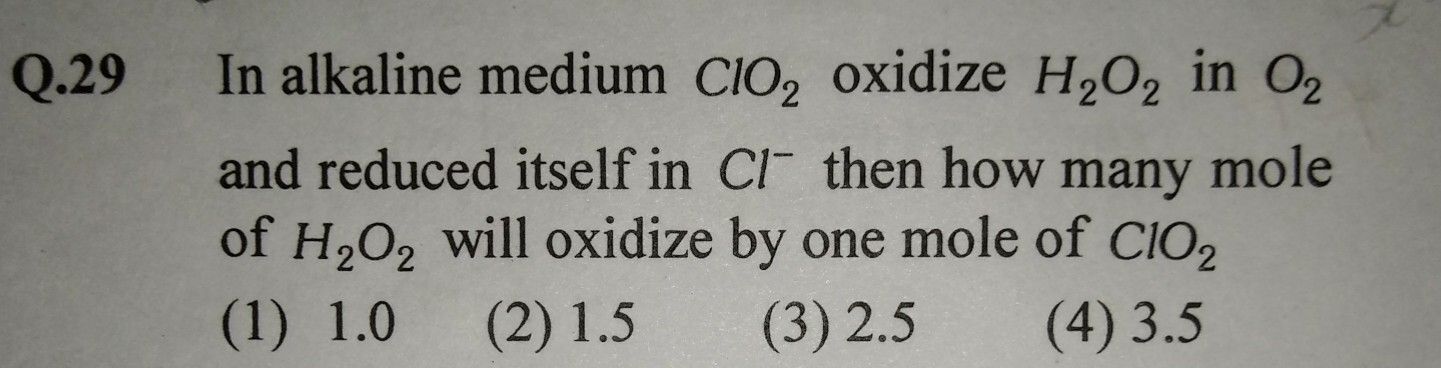
Asked by brijk456 | 13 Aug, 2019, 11:46: PM
The equation for redox reaction is,
ClO2 + H2O2 + OH− → O2 + Cl− + H2O
Half reactions are;
Cl+4 +5e → Cl− 2
2O− − 2e → O20 5
On balancing the equation,
2ClO2 + 5H2O2 + 2OH− → 5O2 + 2Cl− + 6H2O
2 moles of ClO2 oxidise 5 mole of H2O2
So, 1 mole of ClO2 oxidise 2.5 mole of H2O2
Answered by Varsha | 14 Aug, 2019, 04:31: PM
NEET neet - Chemistry
Asked by shubhisingh20001 | 08 Nov, 2023, 01:17: PM
NEET neet - Chemistry
Asked by ssolaimuthu9 | 08 Jul, 2022, 01:01: PM
NEET neet - Chemistry
Asked by rohitraman1115 | 22 May, 2021, 04:13: PM
NEET neet - Chemistry
Asked by arnavvidudala20050 | 17 May, 2020, 03:11: PM
NEET neet - Chemistry
Asked by anjanakurup728 | 21 Nov, 2019, 10:06: AM
NEET neet - Chemistry
Asked by sumayiah2000 | 20 Nov, 2019, 08:09: PM
NEET neet - Chemistry
Asked by Balbir | 28 Aug, 2019, 07:52: PM
NEET neet - Chemistry
Asked by ctmonasara914 | 27 Aug, 2019, 11:01: PM
NEET neet - Chemistry
Asked by brijk456 | 13 Aug, 2019, 11:46: PM