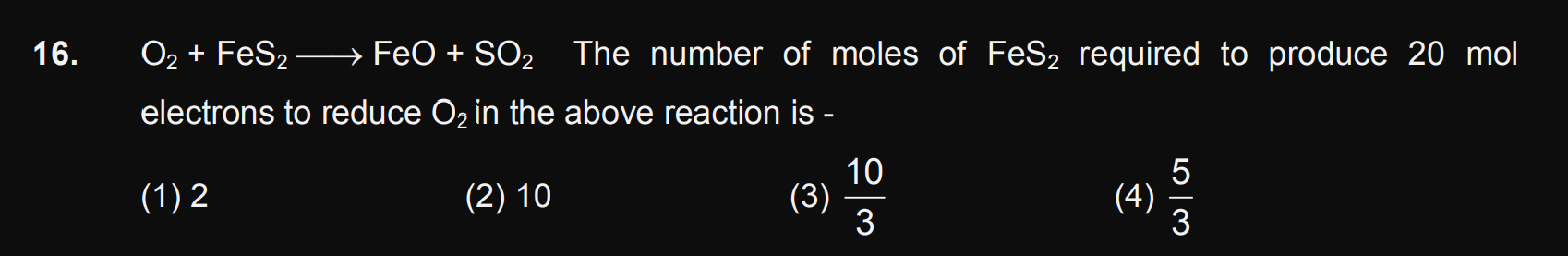JEE Class main Answered
Question no 29
Asked by Adityaaryan681681 | 30 Jul, 2019, 10:15: PM
Given:
Density = 1.15 g/L
P = 740 mm Hg
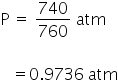
T = 300 K
Using ideal gas equation,
PV = nRT
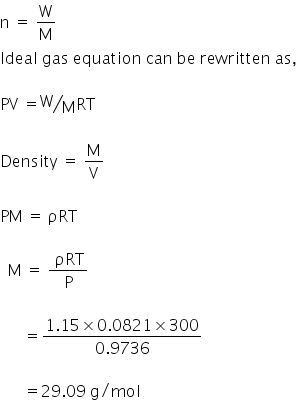
Let % of N2 be X and O2 be (1-X)
Therefore,
28X + (1-X)32 = 29.09
28X + 32- 32X = 29.09
-4X = -2.90
X = 0.7274
=72.74 % of N2
Composition of N2 is 72%
Answered by Varsha | 31 Jul, 2019, 11:01: AM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 05:37: PM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by jadhavshivtej256 | 27 Feb, 2024, 06:25: PM
JEE main - Chemistry
Asked by pradumankumarsah1 | 30 Jan, 2024, 02:36: PM
JEE main - Chemistry
Asked by srujan11042008 | 06 Nov, 2023, 10:31: AM
JEE main - Chemistry
Asked by vuppulojusaritha | 05 Nov, 2023, 02:22: PM
JEE main - Chemistry
Asked by radheshyambaheti085 | 09 Aug, 2023, 07:10: AM
JEE main - Chemistry
Asked by | 17 Aug, 2022, 08:10: PM
JEE main - Chemistry
Asked by aryankatiyar223 | 10 Aug, 2022, 11:57: PM