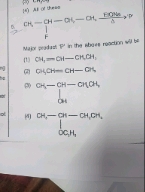
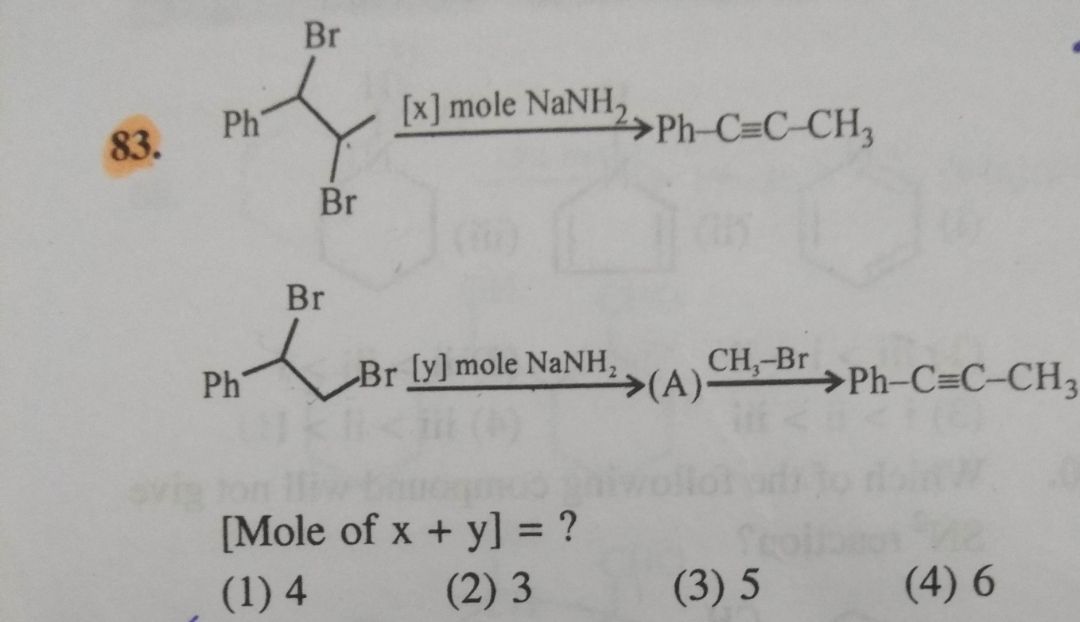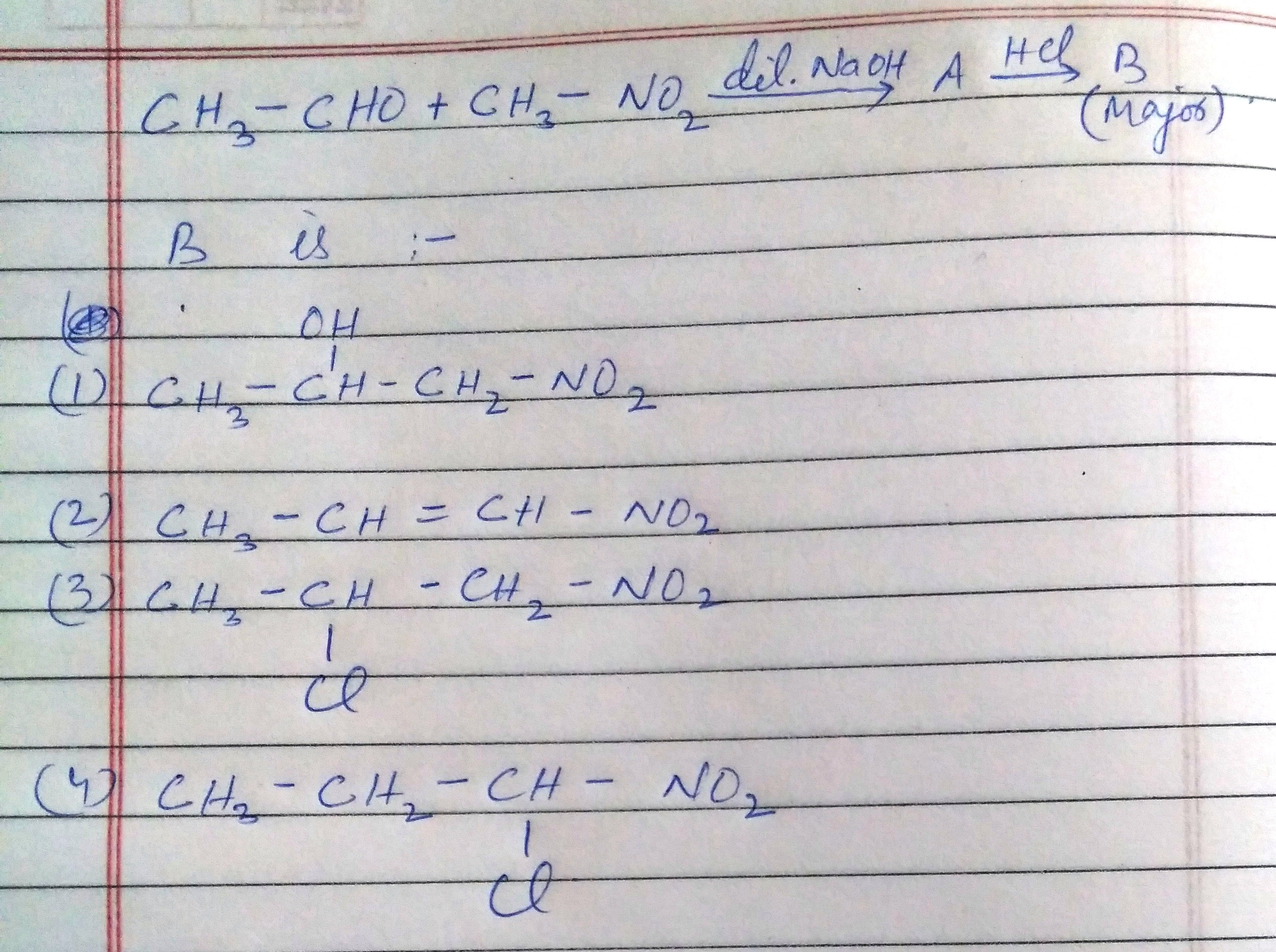NEET Class neet Answered
Please answer the following question with explanation by explaining each option
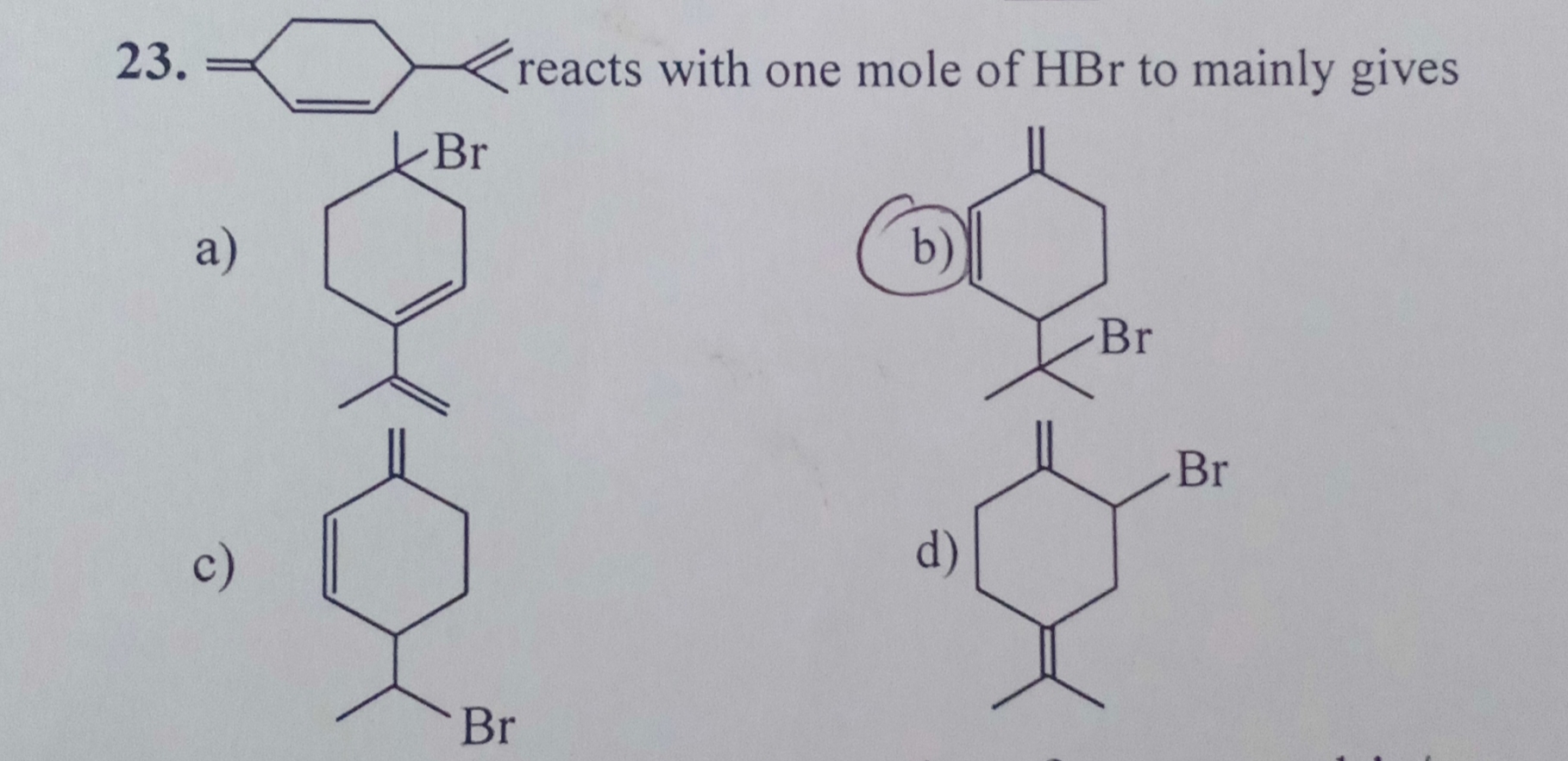
Asked by deepakudgiri29 | 28 Jan, 2019, 06:09: PM
The addition of HBr is carried out by Markovnikov's rule, It predicts the addition of HBr regioselectively to unsymmetrically substituted alkene where X is a halide ion.
According to this rule, halide ion gets added to highly substituted double bonded carbon atom of the alkene.

Hence there are only two possibilities of formation of the product,

Out of these options (a) and (b), option (a) is the correct option because in option (a) the ring strain is released by the formation of carbocation hence reaction proceeds towards less energetic product formation.
The energy of the product should be less than that of the reactant.
Answered by Ramandeep | 29 Jan, 2019, 06:44: PM
NEET neet - Chemistry
Asked by hk62929363 | 04 Mar, 2024, 03:56: AM
NEET neet - Chemistry
Asked by samramojuru | 22 Feb, 2024, 04:18: PM
NEET neet - Chemistry
Asked by swetadayal036 | 15 Jan, 2024, 08:04: PM
NEET neet - Chemistry
Asked by sabhachoudhary0786 | 15 Sep, 2023, 04:53: PM
NEET neet - Chemistry
Asked by jhajuhi19 | 05 Sep, 2020, 05:35: AM
NEET neet - Chemistry
Asked by jhajuhi19 | 11 Aug, 2020, 04:25: AM
NEET neet - Chemistry
Asked by subhasanth | 29 Apr, 2020, 10:53: AM
NEET neet - Chemistry
Asked by jhajuhi19 | 13 Apr, 2020, 08:27: AM
NEET neet - Chemistry
Asked by harunabbas | 08 Apr, 2020, 02:13: PM
NEET neet - Chemistry
Asked by prakriti12oct | 30 Mar, 2020, 01:23: AM