CBSE Class 12-science Answered
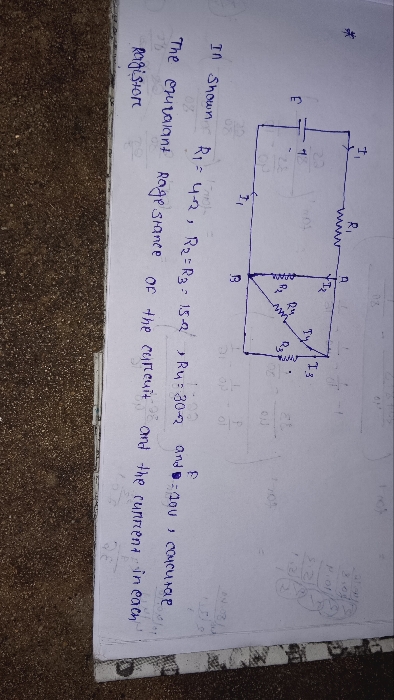
how to find this
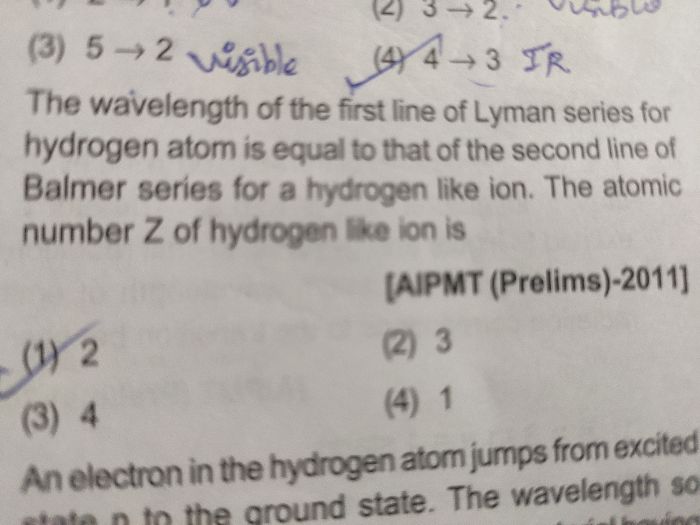
Asked by albiinfanta | 14 Mar, 2020, 08:11: PM
Energy level En of hydrogen like atom for orbital quantum number n is given by
 .......................(1)
.......................(1)First line of Lyman series in hydrogen is due to transition from n=2 to n=1
Hence energy difference for first line of Lyman seriesis in hydrogen atom given by
 .................. (2)
.................. (2)Second line of Balmer series in hydrogen-like atom is due to transition from n=4 to n=2
Hence energy difference for second line of Balmer series in hydrogen-like atom is given by
 ..................(3)
..................(3)By equating equations (2) and (3), we get z = 2
Answered by Thiyagarajan K | 15 Mar, 2020, 09:22: AM
Concept Videos
CBSE 12-science - Physics
Asked by ankush76728 | 06 May, 2024, 04:52: PM
CBSE 12-science - Physics
Asked by ankush76728 | 05 May, 2024, 09:55: PM
CBSE 12-science - Physics
Asked by heymindurownbusiness | 04 May, 2024, 11:15: AM
CBSE 12-science - Physics
Asked by talulu | 01 May, 2024, 05:14: PM
CBSE 12-science - Physics
Asked by kanishkg511 | 30 Apr, 2024, 07:25: PM
CBSE 12-science - Physics
Asked by sahoobanita89 | 30 Apr, 2024, 05:10: AM
CBSE 12-science - Physics
Asked by divakar.9124 | 27 Apr, 2024, 10:42: PM
CBSE 12-science - Physics
Asked by panneer1766 | 24 Apr, 2024, 01:52: PM