CBSE Class 11-science Answered
an ideal gas is enclosed in a cylinder at pressure 2 atm and temperature 300k the mean time between two successive collisions is 6×10–⁸s if the pressure is doubled and temperature is increased to 500k the mean time between two successive collisions will be close to
Asked by ifrayaseen31 | 28 Oct, 2023, 09:26: AM
Initial pressure P = 2 atm ; Final pressure = 5 atm ;
Initial temperature = 300 K ; final temperature = 500 K
Mean free path λ between moleculer collisions is , 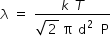
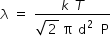
where k is Boltzman constant , T is temperature , d is moleculer size and P is pressure .
Mean free path λ between moleculer collision is directly proportional to temperature T and
inversely proportional to pressure P
Average speed vavg of gas molecules is proportional to square root of temperature ,
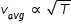
Mean time τ between collision = ( mean free path ) / ( average speed )
( please note that τ in above expression is greek symbol tau not temperature )
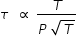
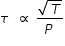
Hence mean time between collision at 500 K and 4 atm. pressure is calculated as
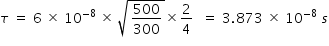
Answered by Thiyagarajan K | 28 Oct, 2023, 11:24: AM
Concept Videos
CBSE 11-science - Physics
Asked by ifrayaseen31 | 28 Oct, 2023, 09:26: AM
CBSE 11-science - Physics
Asked by karanchandra34 | 29 Jan, 2019, 11:10: PM
CBSE 11-science - Physics
Asked by Madhurimaurya609 | 11 Jul, 2018, 08:14: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM