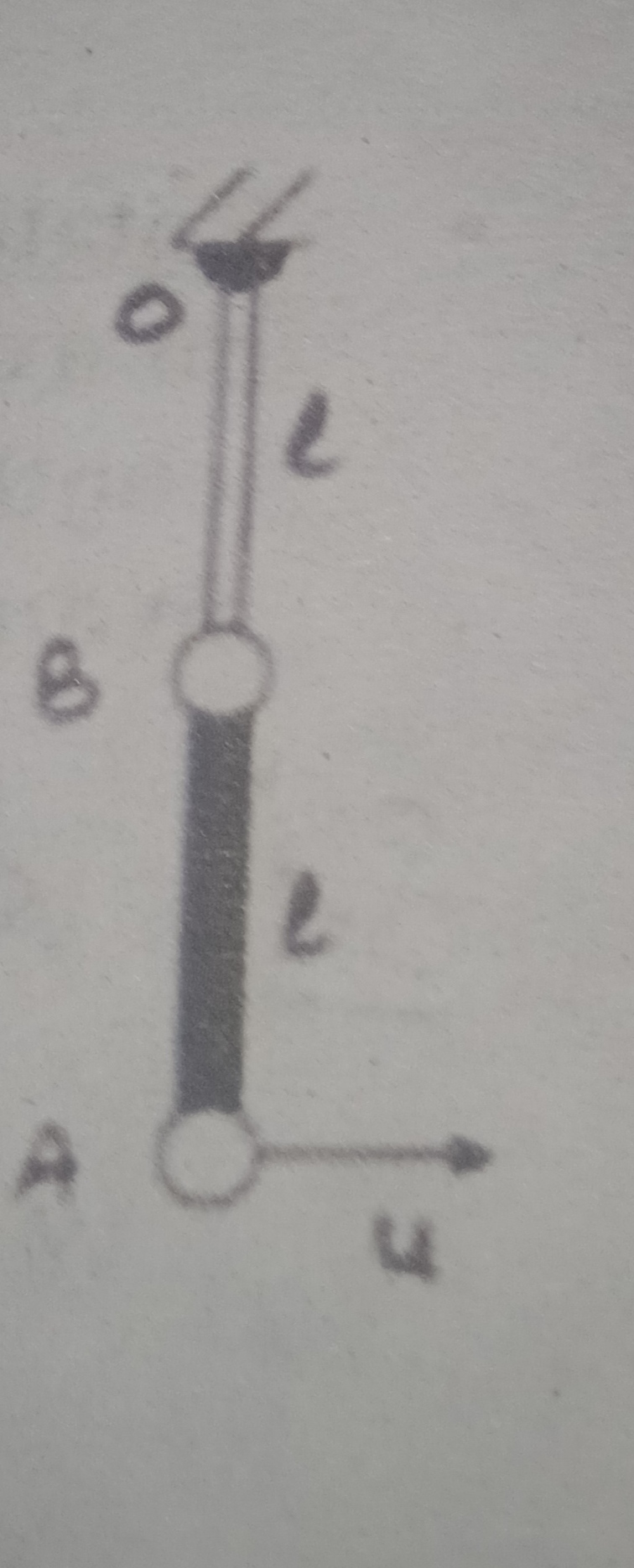CBSE Class 12-science Answered
A vessel contain 100grm of ice at 0 degree celcius . now steam is passed into vessel to melt ice. Neglecting the thermal capacity of the vessel , find the mass of the water in the vessel when all ice melts into water.
Asked by Pankajranagopeshwar | 16 Apr, 2019, 06:36: PM
In question it is given that only ice need to melt so temperature of the water is going to be 0C
Het needed to melt ice = MLf (Lf is the latent heat of fusion)
H = 100 ×80 = 8000 cal
This heat for melting must be provided by steam so mass of steam
H = msΔT
8000 = m× 480×100
So m = 0.16667 kg = 166.67gm
So net mass of water in container = 100+166.67 = 266.7gm
Answered by Ankit K | 16 Apr, 2019, 11:31: PM
Concept Videos
CBSE 12-science - Physics
Asked by artabandhusahu85 | 24 Apr, 2024, 12:07: PM
CBSE 12-science - Physics
Asked by niharvijayvargiya5 | 23 Apr, 2024, 06:40: PM
CBSE 12-science - Physics
Asked by kulhariabhijeet | 21 Apr, 2024, 02:39: PM
CBSE 12-science - Physics
Asked by mohapatraswetalina88 | 21 Apr, 2024, 12:18: PM
CBSE 12-science - Physics
Asked by aishaisha091098 | 19 Apr, 2024, 04:54: PM
CBSE 12-science - Physics
Asked by dasrituparna1999 | 13 Apr, 2024, 06:56: AM
CBSE 12-science - Physics
Asked by dasrituparna1999 | 12 Apr, 2024, 09:26: PM
CBSE 12-science - Physics
Asked by mishrigupta19319 | 08 Apr, 2024, 06:28: PM
CBSE 12-science - Physics
Asked by madhav9119887644 | 07 Apr, 2024, 08:10: PM