CBSE Class 9 Answered
(a)Dry ice is compressed at high pressure. What happens when the pressure is released?
(b)Why is it necessary to cool the gas while applying pressure to liquefy?
Asked by Topperlearning User | 06 May, 2014, 01:36: PM
(a)Dry ice (solid CO2) gets converted into gaseous state on releasing the pressure.
(b)Cooling the gas lowers the temperature of compressed gas and helps in liquefying it.
Answered by | 06 May, 2014, 03:36: PM
Application Videos
Concept Videos
CBSE 9 - Chemistry
Asked by rubinapathan228 | 29 Jun, 2023, 05:45: PM
CBSE 9 - Chemistry
Asked by yforyt3672 | 09 Apr, 2023, 07:37: PM
CBSE 9 - Chemistry
Asked by pranavtamboli65.9 | 02 Aug, 2022, 08:12: PM
CBSE 9 - Chemistry
Asked by ritapriya126 | 20 Apr, 2022, 04:01: PM
CBSE 9 - Chemistry
Asked by renukaramaswamyr | 08 Sep, 2021, 12:32: PM
CBSE 9 - Chemistry
Asked by gpranithasri | 27 Jul, 2021, 01:23: PM
CBSE 9 - Chemistry
Asked by suhanivasi007 | 04 May, 2021, 05:52: PM
CBSE 9 - Chemistry
Asked by pk36229905 | 06 Apr, 2021, 07:38: AM
CBSE 9 - Chemistry
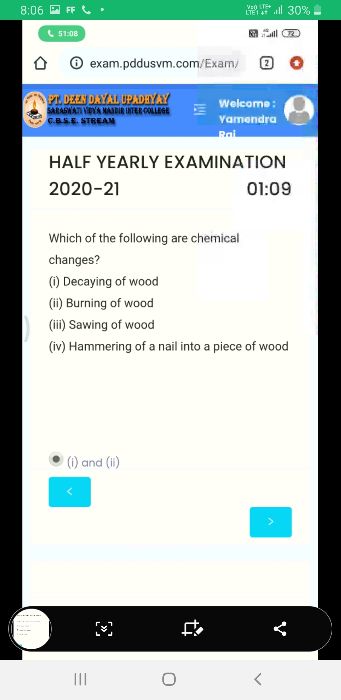
Asked by anubhavev | 30 Oct, 2020, 06:35: PM