CBSE Class 12-science Answered
Please expalin the following question in detail
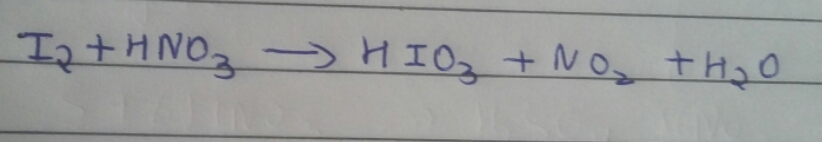
Asked by Balbir | 11 Jan, 2018, 04:03: PM
It’s a simple redox reaction in which oxidation number of nitrogen is decreasing and oxidation number of iodine is increasing.
1. Nitrogen:
O.S. of N in HNO3 is +5 which on reduction become +4
HNO3 → H+ + NO3-
NO3- + 2H+ + e- → NO2 + H2O
i.e., N+5 + e- → N+4 ………………(1)
2. Iodine:
O.S. of I in I2 is 0 which on oxidation become +5
I2 + 6H2O → 2IO3- + 12 H+ + 10 e-
i.e., I20 → 2 I+5 + 10 e- …………..(2)
Multiply eq (1) by 10, we get,
10N+5 + 10e- → 10 N+4 ….(3)
By adding eq(2) by (3) we get,
10N+5 + I20 → 2 I+5 + 10 N+4
Thus the original form of equation is written as,
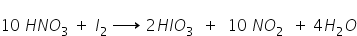
Answered by Ramandeep | 11 Jan, 2018, 06:55: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by shwetayaligar205 | 21 Dec, 2022, 07:45: PM
CBSE 12-science - Chemistry
Asked by kushwaharitik9129 | 14 Jul, 2022, 01:06: PM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 17 Apr, 2020, 09:22: AM
CBSE 12-science - Chemistry
Asked by adarshkamble130 | 19 Aug, 2019, 12:22: AM
CBSE 12-science - Chemistry
Asked by ranvirsingh1as | 16 Dec, 2018, 10:50: PM
CBSE 12-science - Chemistry
Asked by ranvirsingh1as | 16 Dec, 2018, 10:47: PM
CBSE 12-science - Chemistry
Asked by Sneha | 16 Dec, 2018, 03:14: PM
CBSE 12-science - Chemistry
Asked by lekhakarthikeyan | 28 Aug, 2018, 03:54: AM
CBSE 12-science - Chemistry
Asked by lekhakarthikeyan | 28 Aug, 2018, 03:52: AM
CBSE 12-science - Chemistry
Asked by niharikapabba2605 | 07 Aug, 2018, 01:00: AM





 to produce a brown coloured gass which intensifies on addition on copper turnings . on adding dilute ferrous sulphate olution to an aqueous solution to an aquaeous solution of X and then carefully adding conc.
to produce a brown coloured gass which intensifies on addition on copper turnings . on adding dilute ferrous sulphate olution to an aqueous solution to an aquaeous solution of X and then carefully adding conc. 