CBSE Class 7 Answered
Write a word equation to represent the process of rusting of iron.Also write the chemical symbols and formulae of all the substances involved
Asked by uddu1112 | 18 May, 2019, 09:01: AM
Rusting is a slow oxidation process in which iron slowly reacts with oxygen of the air in the presence of moisture and produces a flaky brown substance called rust.
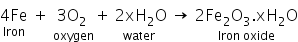
Rust is hydrated ferric oxide ((Fe2O3.xH2O) which forms a reddish brown coating over iron.
Answered by Ramandeep | 18 May, 2019, 10:13: AM
Concept Videos
CBSE 7 - Science
Asked by tkrajkumar87 | 20 Aug, 2023, 10:20: AM
CBSE 7 - Science
Asked by smtchetan | 17 Aug, 2020, 11:57: AM
CBSE 7 - Science
Asked by ezeeswati | 11 Sep, 2019, 18:36: PM
CBSE 7 - Science
Asked by Parshotamlal04 | 08 Jul, 2019, 16:46: PM
CBSE 7 - Science
Asked by uddu1112 | 18 May, 2019, 09:01: AM
CBSE 7 - Science
Asked by sm_tomar | 10 Aug, 2018, 19:49: PM
CBSE 7 - Science
Asked by Topperlearning User | 29 Apr, 2014, 14:42: PM
CBSE 7 - Science
Asked by Topperlearning User | 29 Apr, 2014, 14:34: PM


